NEUROTOXICOLOGY.
NEUROTOXICITY CHEMICAL SAFETY.
DE-RISK FUNCTIONAL ALTERATIONS EFFECTS BEFORE THE CLINIC.
Benefits.
By providing human-relevant, compartmentalized neuronal readouts of compound-induced neurotoxicity , NETRI Neurotoxicity chemical safety platform strengthens clinical risk assessment. It enables early detection of functional alterations and dose-dependent effects, supporting safer chemical or pesticide evaluation, guiding regulatory decisions, and reducing reliance on animal testing.
Description.
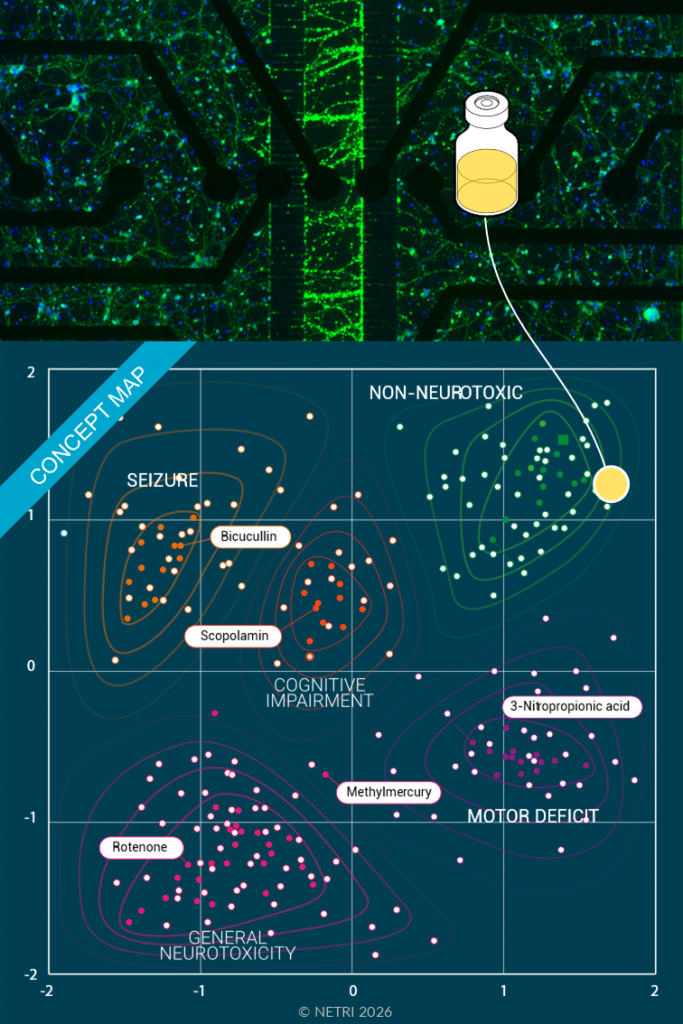
Create a Gluta-Gluta co-culture but only subject one compartment to the compound to be tested. This allows to test compound’s neurotoxicity against a chemical or pesticide references that acts as the platform’s Neurons as a Sensor.
Readouts.
• MEA recordings.
• Digital signatures.
• Imaging.
Advantages.
• Direct recording of neurotoxic activity alteration.
• Possibility to monitor over time.
• Currently under assessment by French ANSES.
OUR OFFERS.
Initialization.
Creation of NaaS biology platform and NaaS Model for a target application.
• In Your Lab: Develop your NaaS Model on your own terms.
• At NETRI: Let NETRI Develop your NaaS Model.
Upscaling.
Build up of Digital NaaS model through Training Library of 100s of reference compounds.
• Pre-Licensing: Test your molecules using your NaaS Model.
Screening & Drug Design.
Transfer of methodology, devices and predictive model capabilities to client.
• Licensing: Screen your molecules using an existing NaaS Model.
Regulatory Approval.
Extension of approved NaaS Model to multiple clients. Larger deals with CROs to handle volumes.
• Licensing & milestones.