WETWARE.
WETWARE APPROACH.
At NETRI, we have developed and validated a comprehensive portfolio of human-relevant cell models specifically optimized for our organs on chip platforms and associated standardized operating protocols. Our wetware approach integrates a wide range of cell types, each functionally qualified within our devices through standardized workflows combining advanced culture conditions, compartmentalization, readouts (imaging, dosing, electrophysiological…).
We have successfully validated the integration and performance of five major cell categories within our systems: Neuronal Cells, Neuronal Associated Cells, Immune Cells, Other Cell Types, and 3D Cell Cultures. Each category has been rigorously tested to ensure compatibility with our platforms, reproducibility of results, and relevance for translational applications.
This unique combination of optimized cell sources and device-specific protocols enables the creation of complex, physiologically relevant human models, supporting a broad range of applications from fundamental research to drug discovery, safety assessment, and advanced functional diagnostics.
NEURONAL CELLS.
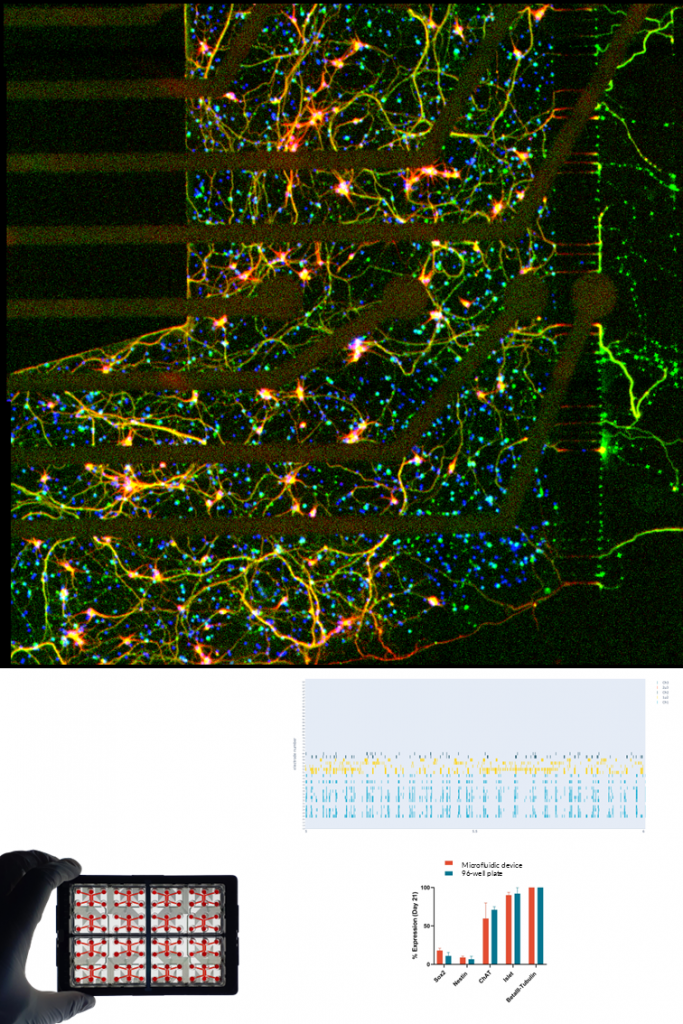
SENSORY NEURONS.
Human iPSC-derived sensory neurons exhibit robust expression of key nociceptive markers (e.g., TRPV1, Nav1.7, Nav1.8) and develop dense, polarized neurite networks within NETRI platforms. Functional validation demonstrates their ability to respond to a wide range of stimuli, including inflammatory mediators (e.g., TNF-α), chemotherapeutic agents (e.g., paclitaxel), and physical such as temperature and osmotic changes.
Key applications include:
• Pain modeling (nociceptive and inflammatory pathways).
• Chemotherapy-Induced Peripheral Neuropathy (CIPN).
• Drug screening targeting ion channels and receptors.
• AI-driven phenotypic profiling and signature generation.
Available suppliers: Anatomic ; Axol Bioscience ; bit.bio.
MOTOR NEURONS.
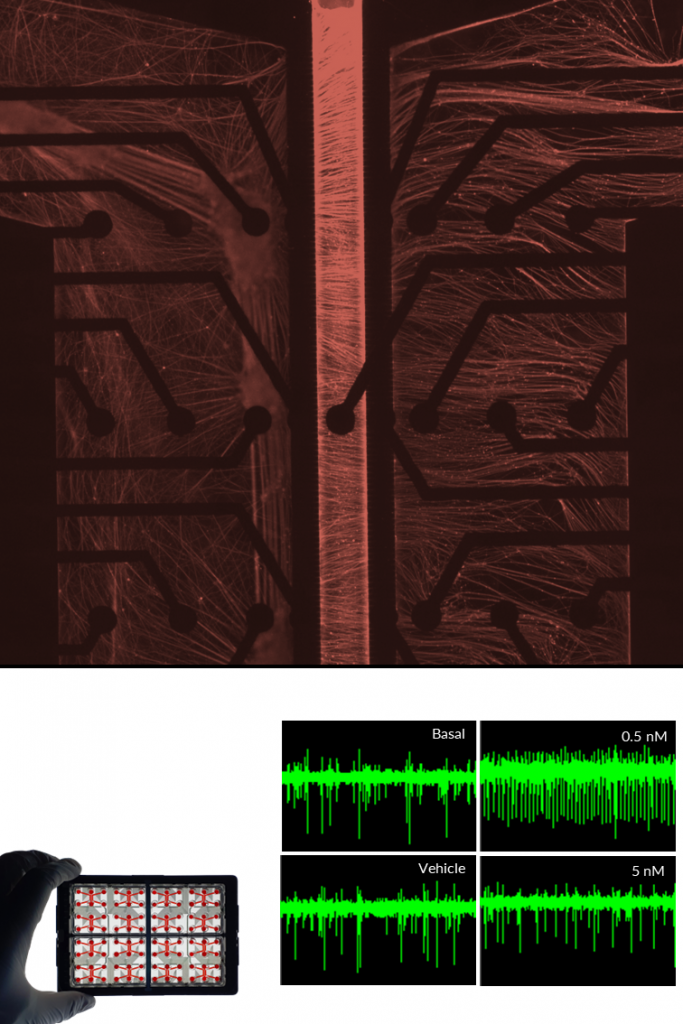
Human iPSC-derived motor neurons form mature neuronal networks characterized by extensive axonal projections and synaptic connectivity. Their electrophysiological activity can be reliably recorded using MEA, demonstrating spontaneous firing and network synchronization over time.
In compartmentalized systems, motor neurons enable precise modeling of axonal growth, injury, and regeneration processes.
Key applications include:
• Neurodegenerative disease modeling (e.g., ALS).
• Axonal injury and regeneration studies.
• Neuromuscular junction (NMJ) modeling (in co-culture).
• Drug screening for neuroprotection and regeneration.
Available suppliers: Anatomic ; Axol Bioscience ; bit.bio ; FUJIFILM Cellular Dynamics.
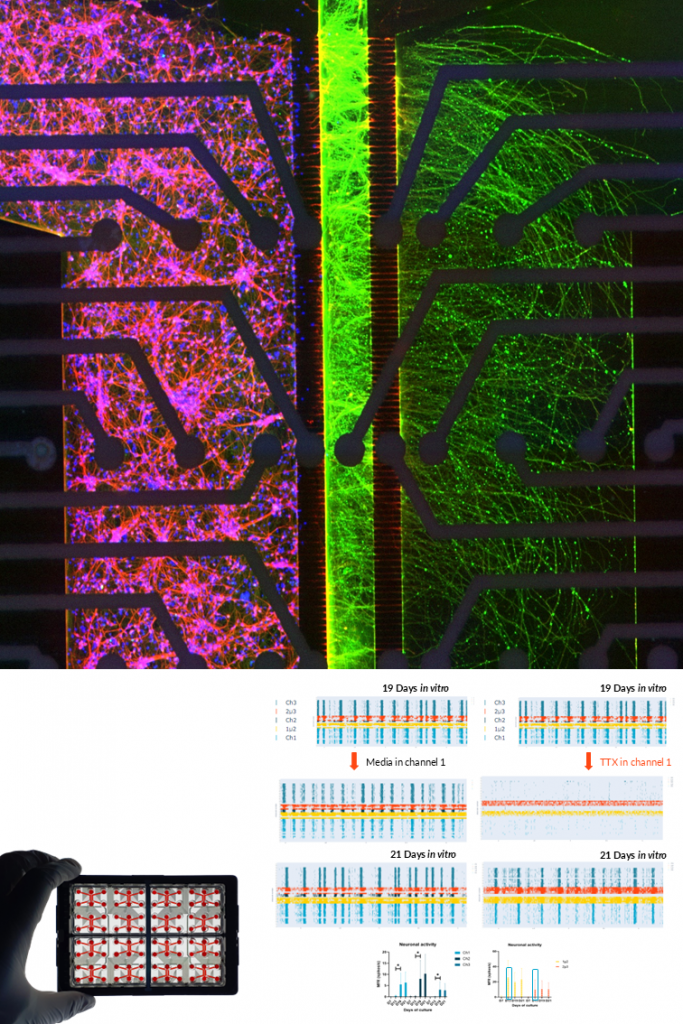
GLUTAMATERGIC NEURONS.
Human iPSC-derived glutamatergic neurons form active, synchronized networks with strong electrophysiological signatures measurable via MEA. Their activity can be modulated pharmacologically (e.g., TTX inhibition), confirming functional maturity.
Additionally, co-culture with tumor cells (e.g., pediatric glioblastoma) demonstrates their relevance in modeling neuron–tumor interactions and co-culture with patient-CSF demonstrates their relevance in Neurodegenerative disease diagnosis.
Key applications include:
• Neurodegenerative diseases diagnosis (e.g., AD).
• Neuro-oncology (tumor–neuron interactions).
• Synaptic activity and network dynamics studies.
• Drug screening targeting excitatory transmission.
• Disease modeling (e.g., epilepsy, neurodegeneration).
Available suppliers: BrainXell ; FUJIFILM Cellular Dynamics.
GABAERGIC NEURONS.
Human iPSC-derived GABAergic neurons provide inhibitory control within neuronal networks and are essential for modeling the excitation/inhibition (E/I) balance. Within NETRI platforms, they integrate into functional circuits and modulate network activity, enabling the study of synaptic regulation and neuronal oscillations.
Key applications include:
• Epilepsy and seizure disorder modeling.
• Neurodevelopmental disorders (e.g., ASD).
• Synaptic pharmacology and network modulation.
• E/I balance assessment in complex co-culture systems.
Available suppliers: BrainXell.
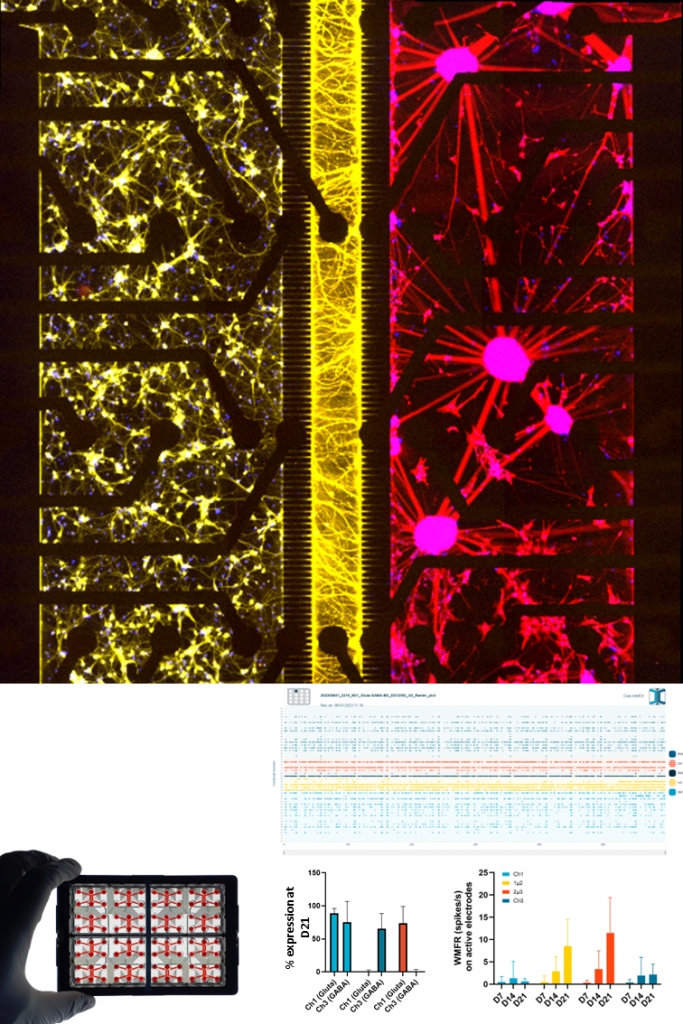
DOPAMINERGIC NEURONS.
Human iPSC-derived dopaminergic neurons characterized by the expression of tyrosine hydroxylase (TH) and dopamine transporters, are critical for modeling reward, motor control, and neurodegenerative diseases. Within NETRI platforms, they develop functional networks and can be used to assess degeneration, connectivity loss, and pharmacological modulation.
Key applications include:
• Parkinson’s disease modeling.
• Neurotoxicity and neuroprotection studies.
• Dopamine-related pathway screening.
• Long-range connectivity and degeneration assays.
Available suppliers: FUJIFILM Cellular Dynamics.
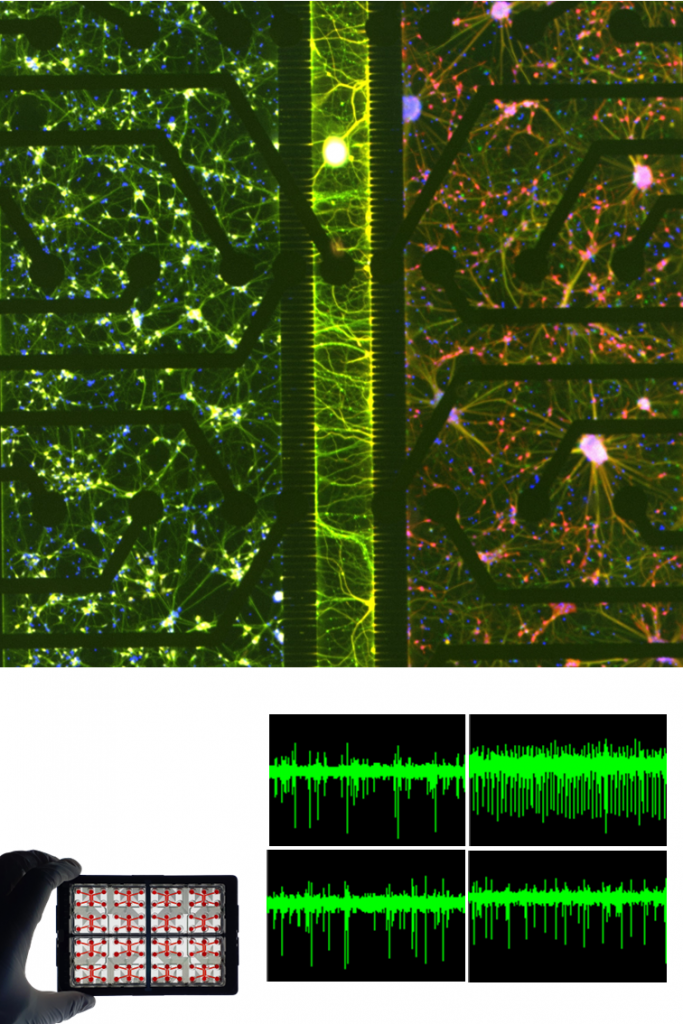
NEURONAL ASSOCIATED CELLS.
ASTROCYTES.
Human astrocytes are key regulators of neuronal homeostasis, synaptic function, and metabolic support. Within NETRI platforms, astrocytes co-cultured with neurons (e.g., sensory neurons) significantly enhance network maturation, stability, and responsiveness.
Their presence modulates neuronal excitability and contributes to more physiologically relevant signal propagation, as demonstrated by increased synchronization and more robust electrophysiological activity in MEA recordings. Astrocytes also play a central role in neuroinflammation and neurodegenerative processes, making them indispensable for disease modeling.
Key applications include:
• Neuron–glia interaction studies.
• Neuroinflammation and neurodegeneration modeling (e.g., Alzheimer’s disease).
• Enhancement of neuronal maturation and network stability.
• Functional assay development with improved physiological relevance.
• Drug screening targeting neuro-glial interactions.
Available suppliers: ScienCell ; Thermo Fisher Scientific ; Lonza.
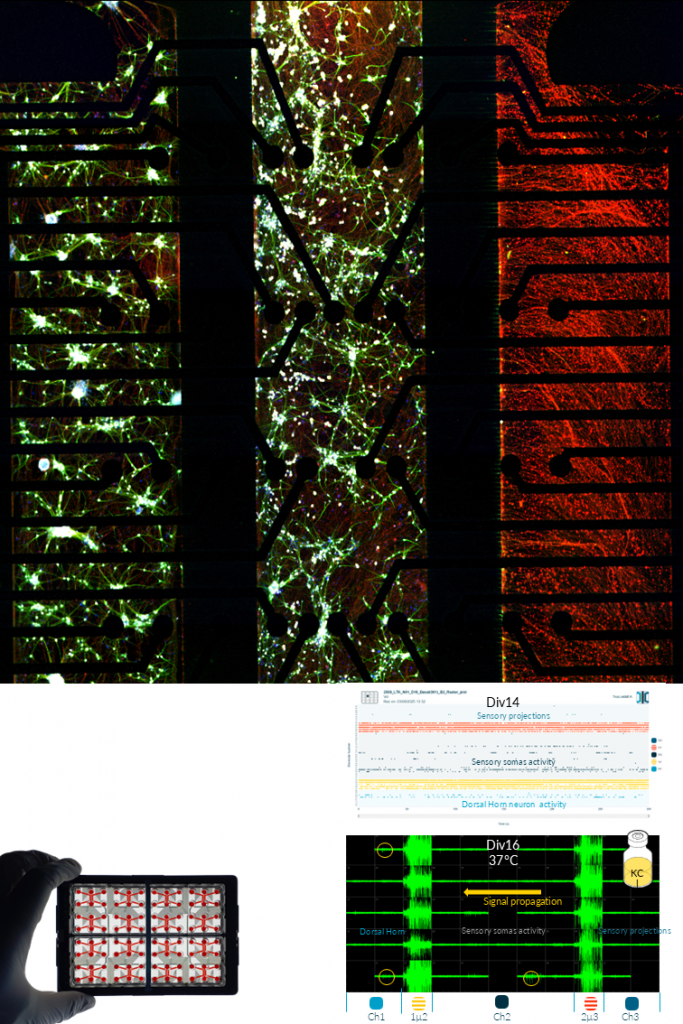
SCHWANN CELLS.
Schwann cells are the principal glial cells of the peripheral nervous system and are essential for axonal support, myelination, and regeneration. Within NETRI’s compartmentalized platforms, they enable the recreation of peripheral nerve-like architectures, supporting long-range axonal growth and functional connectivity.
Their integration into co-culture systems with sensory or motor neurons allows precise modeling of axon–glia interactions, including myelination processes and response to injury. Schwann cells also play a key role in nerve repair mechanisms, making them highly relevant for regeneration and neurotoxicity studies.
Key applications include:
• Peripheral nerve modeling and myelination studies.
• Axonal injury and regeneration assays.
• Chemotherapy-induced peripheral neuropathy (CIPN) modeling.
• Neurotoxicity and neuroprotection screening.
• Functional reconstruction of peripheral neural circuits.
Available suppliers: ScienCell ; Lonza.
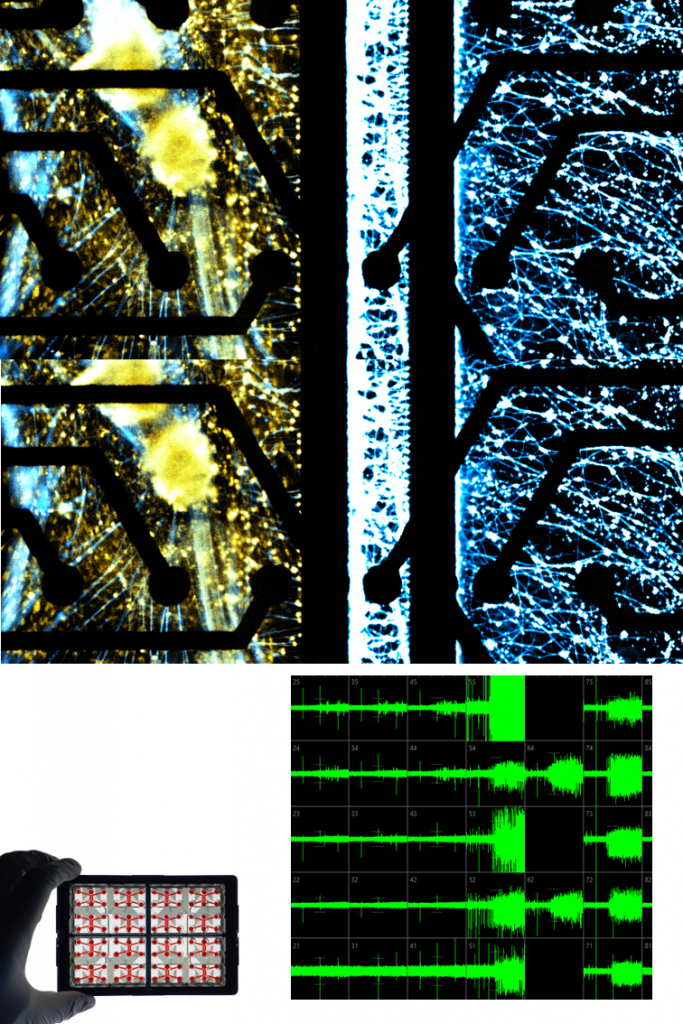
IMMUNE CELLS.
MACROPHAGES.
Macrophages play a central role in innate immunity, inflammation, and tissue remodeling. Within NETRI platforms, they demonstrate active migration, cytokine release, and interaction with neuronal compartments, enabling the study of localized inflammatory responses.
Their integration allows modeling of immune cell infiltration and its impact on neuronal function, including modulation of electrophysiological activity and network integrity.
Key applications include:
• Neuroinflammation modeling.
• Immune cell recruitment and migration assays.
• Cytokine-mediated neuronal modulation.
• Drug screening for anti-inflammatory compounds.
• Peripheral neuropathy and injury models.
Available suppliers: Lonza ; Thermo Fisher Scientific ; STEMCELL Technologies.

PERIPHERAL BLOOD MONONUCLEAR CELLS (PBMCs).
PBMCs represent a heterogeneous population of immune cells (including lymphocytes and monocytes) and provide a clinically relevant model for studying systemic immune responses. In NETRI platforms, PBMCs can be introduced into specific compartments to investigate their interactions with neuronal networks and their response to external stimuli.
Their use enables the recreation of complex immune environments and supports patient-derived approaches for personalized medicine.
Key applications include:
• Patient-specific immune response modeling.
• Neuro–immune interaction studies.
• Cytokine profiling and inflammatory response assessment.
• Drug screening and immunotoxicity testing.
• Translational and personalized medicine approaches.
Available suppliers: Lonza ; AllCells ; STEMCELL Technologies.
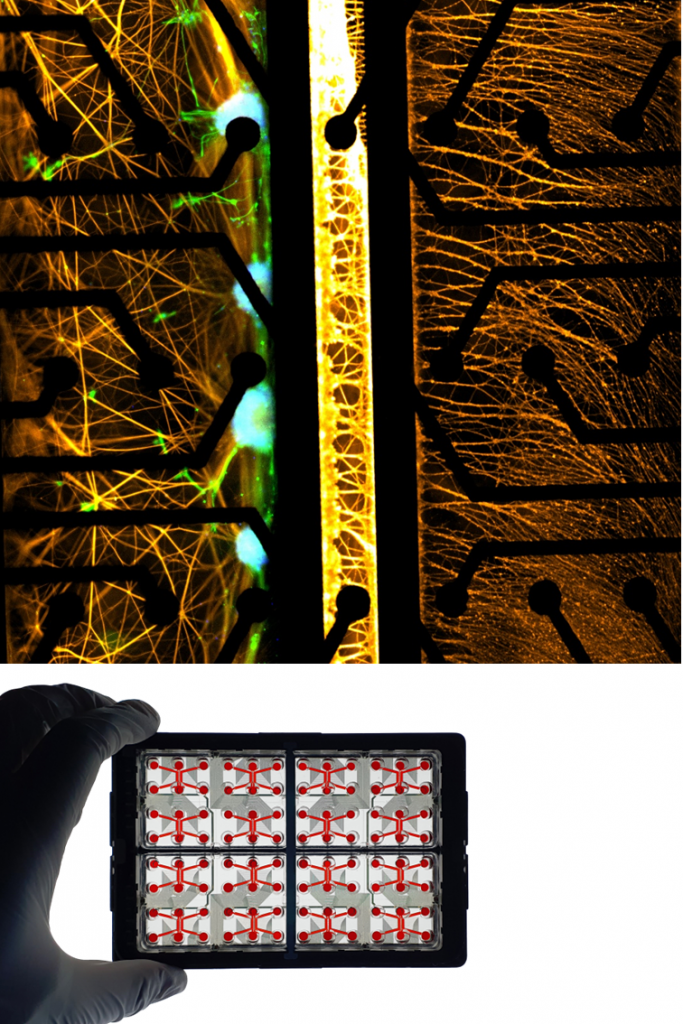
LYMPHOCYTES.
Lymphocytes, including T cells and B cells, are key actors of the adaptive immune response. Within NETRI platforms, they enable the study of antigen-specific responses and their impact on neuronal function.
Their interaction with neuronal and glial cells provides insights into chronic inflammation, autoimmune mechanisms, and immune-mediated neurotoxicity.
Key applications include:
• Adaptive immune response modeling.
• Autoimmune disease studies (e.g., multiple sclerosis).
• Neuroinflammation and chronic inflammation models.
• Immuno-oncology applications.
• Evaluation of immunotherapies.
Available suppliers: STEMCELL Technologies ; Miltenyi Biotec.

DENDRITIC CELLS.
Dendritic cells are professional antigen-presenting cells (APCs) that play a pivotal role in initiating and regulating adaptive immune responses. Within NETRI platforms, dendritic cells can be integrated into compartmentalized systems to model antigen capture, processing, and presentation, as well as their interactions with both neuronal and immune compartments.
Functionally, dendritic cells are validated in our devices through their ability to respond to inflammatory stimuli, undergo maturation (e.g., upregulation of CD80/CD86 and MHC-II), and secrete cytokines that influence surrounding cell populations. Their presence enables the study of immune activation cascades and their downstream effects on neuronal activity and network behavior.
Key applications include:
• Antigen presentation and immune activation studies.
• Neuro–immune interface modeling.
• Immunotherapy evaluation.
• Cytokine-driven modulation of neuronal activity.
• Chronic inflammation and autoimmune disease modeling.
Available suppliers: STEMCELL Technologies ; Miltenyi Biotec ; Lonza.

OTHER CELL TYPES.
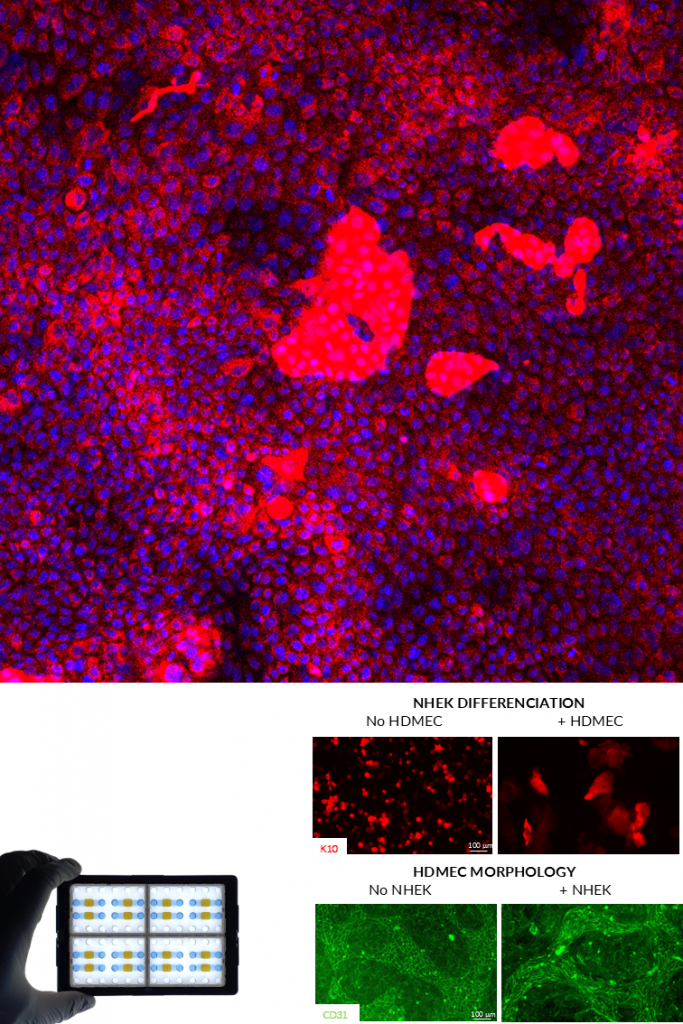
KERATINOCYTES .
Primary human keratinocytes are essential for modeling the epidermal barrier and its interaction with sensory nerve endings. Within NETRI platforms, they form structured epithelial layers and can be co-cultured with sensory neurons to recreate skin–nerve interfaces (innervated skin) or with endothelial cells to recreate vascularized skin.
These systems enable the study of cutaneous responses to chemical, mechanical, and inflammatory stimuli, as well as neuron-mediated signaling in the skin.
Key applications include:
• Skin innervation modeling.
• Irritation and sensitization assays.
• Neurogenic inflammation studies.
• Cosmetic and dermatological testing.
• Peripheral sensory response profiling.
Available suppliers: PromoCell.
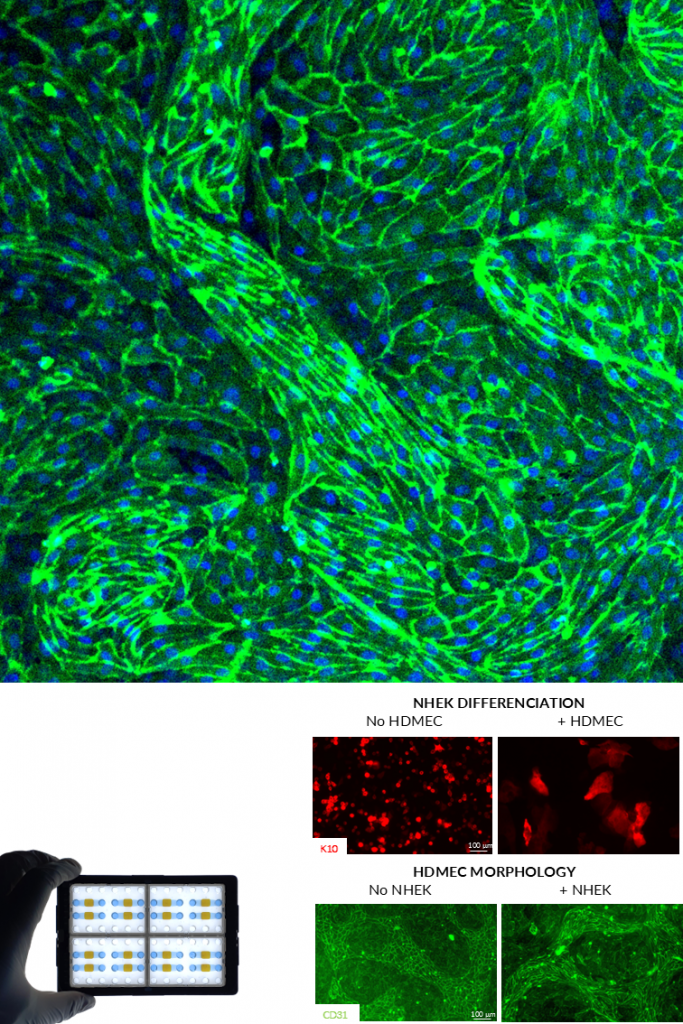
ENDOTHELIAL CELLS.
Endothelial cells are key components of vascular barriers and play a crucial role in regulating permeability, inflammation, and cell trafficking. In NETRI platforms, they can be used to recreate vascular-like interfaces, including simplified blood–brain barrier (BBB) or peripheral vascular environments or vascularized skin.
Functionally, endothelial cells demonstrate barrier formation, selective permeability, and responsiveness to inflammatory signals, enabling the study of molecular transport and immune cell transmigration.
Key applications include:
• Blood–brain barrier (BBB) and vascular modeling.
• Permeability and transport studies.
• Inflammation and immune cell transmigration assays.
• Drug delivery and pharmacokinetics evaluation.
• Neurovascular interaction studies.
Available suppliers: Lonza ; PromoCell ; Thermo Fisher Scientific.
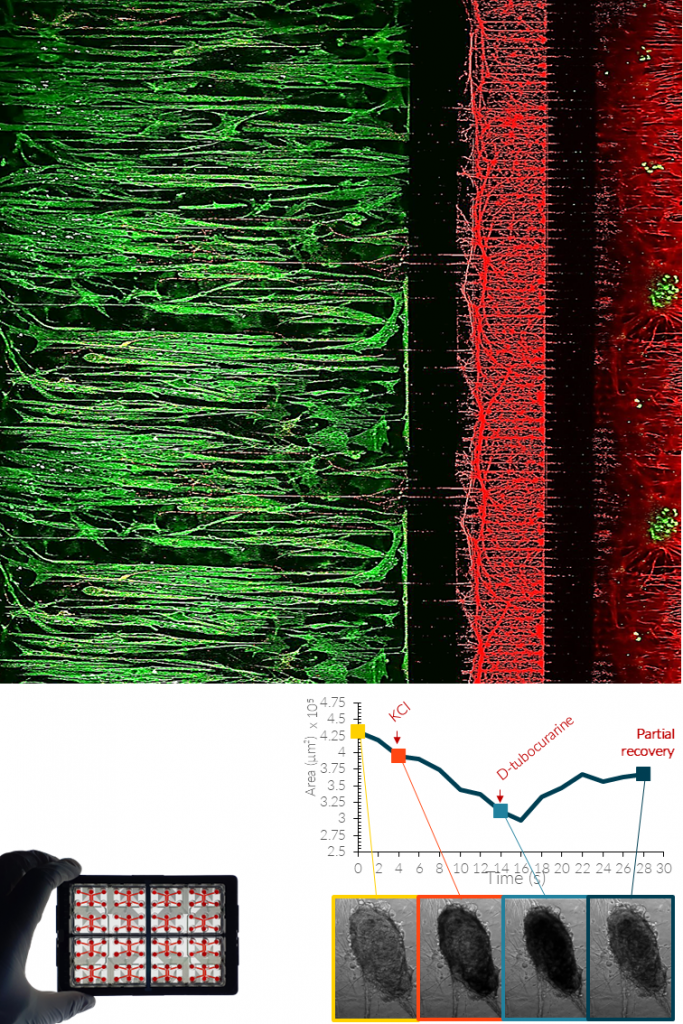
MUSCLE CELLS.
Skeletal muscle cells can be integrated into NETRI platforms to recreate functional neuromuscular interfaces. When co-cultured with motor neurons, they enable the formation of neuromuscular junction (NMJ)-like structures and functional coupling between neuronal activity and muscle contraction.
These systems allow direct assessment of neuromuscular function, degeneration, and response to pharmacological compounds.
Key applications include:
• Neuromuscular junction (NMJ) modeling.
• Muscle contraction and functional assays.
• Neurodegenerative disease modeling (e.g., ALS).
• Drug screening targeting neuromuscular function.
• Toxicity assessment on muscle tissue.
Available suppliers: bit.bio ; Axol Bioscience.
CARDIAC CELLS.
Human cardiomyocytes provide a robust model for assessing cardiac electrophysiology and contractility. Within NETRI-compatible systems, they exhibit spontaneous beating activity and can be functionally characterized through electrophysiological recordings and imaging.
Their integration enables the study of cardio-neural interactions, systemic toxicity, and off-target drug effects.
Key applications include:
• Cardiotoxicity assessment.
• Electrophysiological and contractility studies.
• Multi-organ interaction modeling (neuro–cardiac axis).
• Drug safety pharmacology.
• Predictive toxicology.
Available suppliers: FUJIFILM Cellular Dynamics ; Lonza.
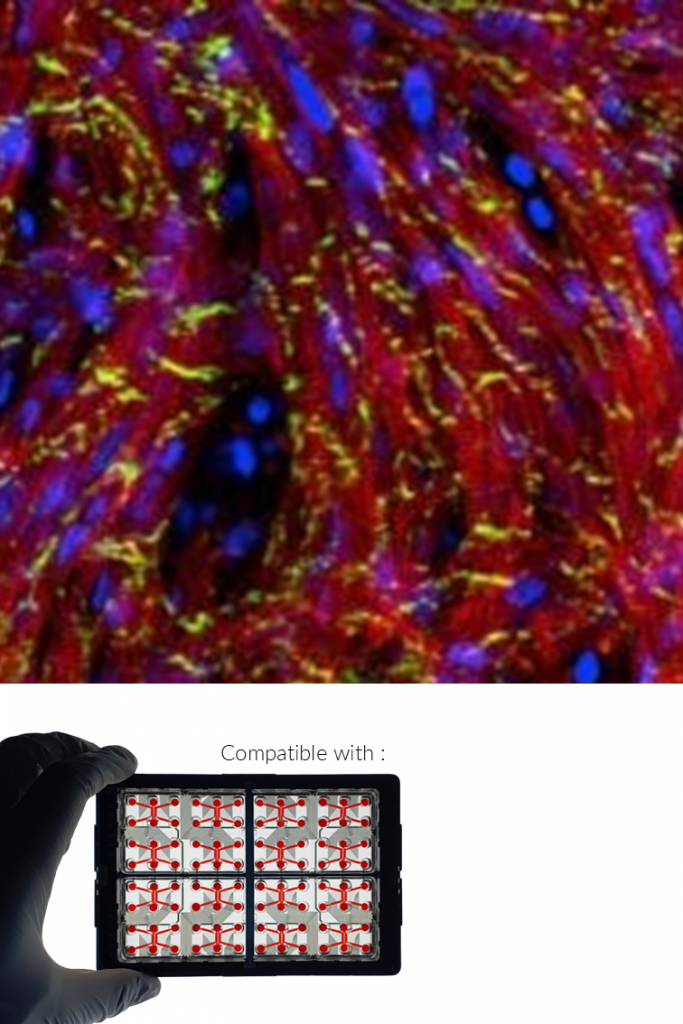
3D CELL CULTURES.
LIVER ORGANOIDS.
BioMIMESYS® supports the generation of mature and functional liver organoids from human cells, including hepatocytes, stellate cells, cholangiocytes, and liver sinusoidal endothelial cells (LSECs). In contrast to 2D cultures, these 3D systems exhibit enhanced differentiation, structural organization, and metabolic activity.
Functional validation includes: increased albumin secretion over time, active CYP3A4 metabolism, demonstrating drug-processing capability and presence of multiple liver cell types and tissue-like architecture.
Key applications include:
• Drug metabolism and hepatotoxicity studies.
• Liver disease modeling.
• Organoid-based screening platforms.
• Long-term functional assays.
Available suppliers: BioMIMESYS® LIVER - ECM hydrogel.
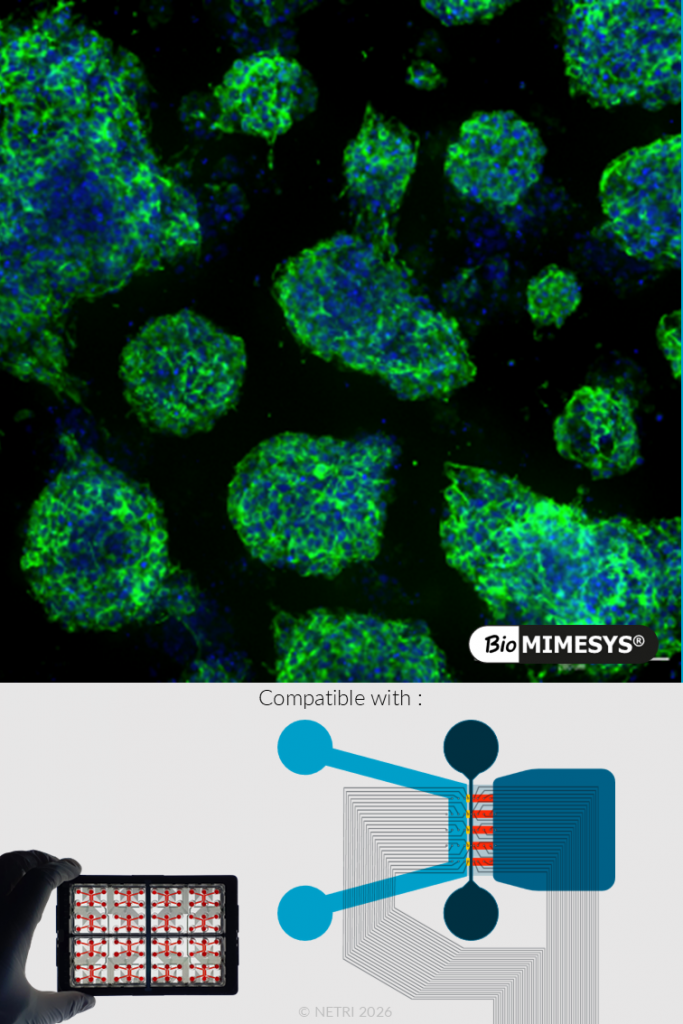
PANCREATIC ORGANOIDS.
Pancreatic tumor spheroids (e.g., PANC-1) cultured in BioMIMESYS® demonstrate controlled growth kinetics and sustained metabolic activity over time. The system supports the formation of reproducible and scalable 3D spheroids.
Functional validation includes: Progressive increase in spheroid size over time, Sustained metabolic activity (WST-1 assay), stable long-term culture in 3D environment.
Key applications include:
• Pancreatic cancer modeling.
• Tumor growth and proliferation studies.
• Drug efficacy and resistance testing.
• High-throughput screening in 3D.
Available suppliers: BioMIMESYS® PANCREACTIC - ECM hydrogel.

ADIPOSE TISSUE ORGANOIDS.
BioMIMESYS® enables the differentiation and maturation of human primary pre-adipocytes into functional adipose tissue. Compared to 2D conditions, cells cultured in 3D show significantly enhanced lipid accumulation and long-term viability.
Functional validation includes: increase triglyceride (TG) accumulation over time over time, improved cell survival and long-term culture stability , enhanced morphological organization of adipocytes.
Key applications include:
• Metabolic disease modeling (e.g., obesity, diabetes).
• Lipid metabolism studies.
• Drug screening targeting adipose tissue.
• Long-term metabolic assays.
Available suppliers: BioMIMESYS® ADIPOSE TISSUE - ECM hydrogel.
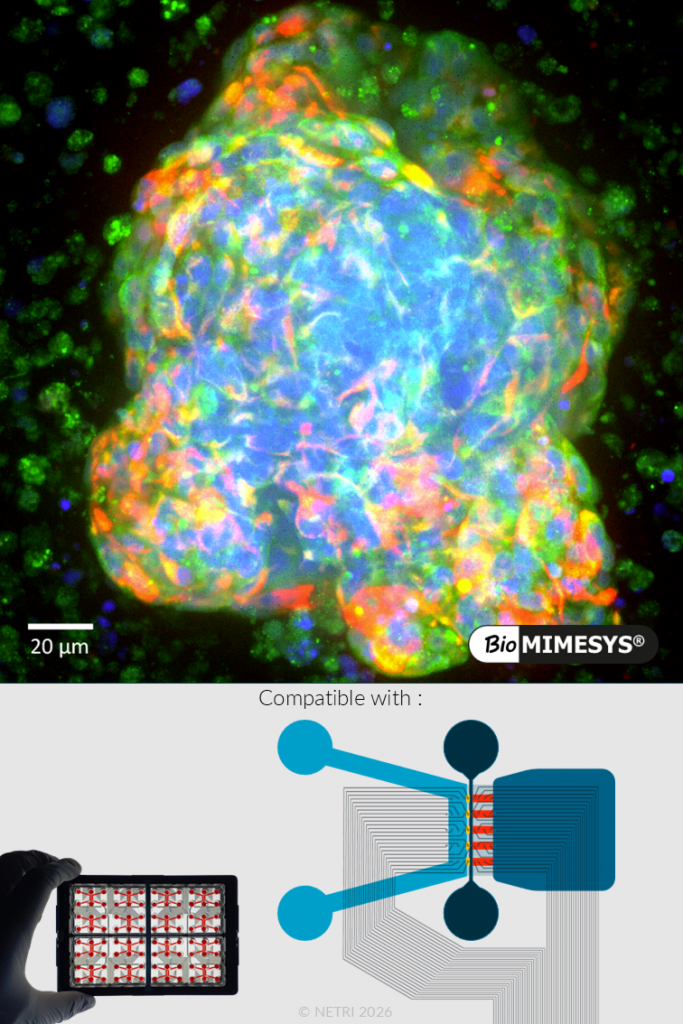
BRAIN ORGANOIDS.
BioMIMESYS® supports the formation of brain organoids and neuronal networks with enhanced structural and functional maturation. These systems exhibit expression of key neuronal markers (e.g., β3-tubulin) and develop complex 3D architectures. Additionally, the tunable mechanical properties of BioMIMESYS® enable adaptation to brain-like environments, supporting more physiologically relevant models.
Functional validation includes: formation of neurospheres and neuronal networks, Expression of neuronal markers and structural organization, Time-dependent maturation (7–21 days progression).
Key applications include:
• Neurodevelopmental and neurodegenerative disease modeling.
• Neural network formation and connectivity studies.
• Drug screening and neurotoxicity assessment.
• Integration with MEA for functional readouts.
Available suppliers: BioMIMESYS® BRAIN - ECM hydrogel.
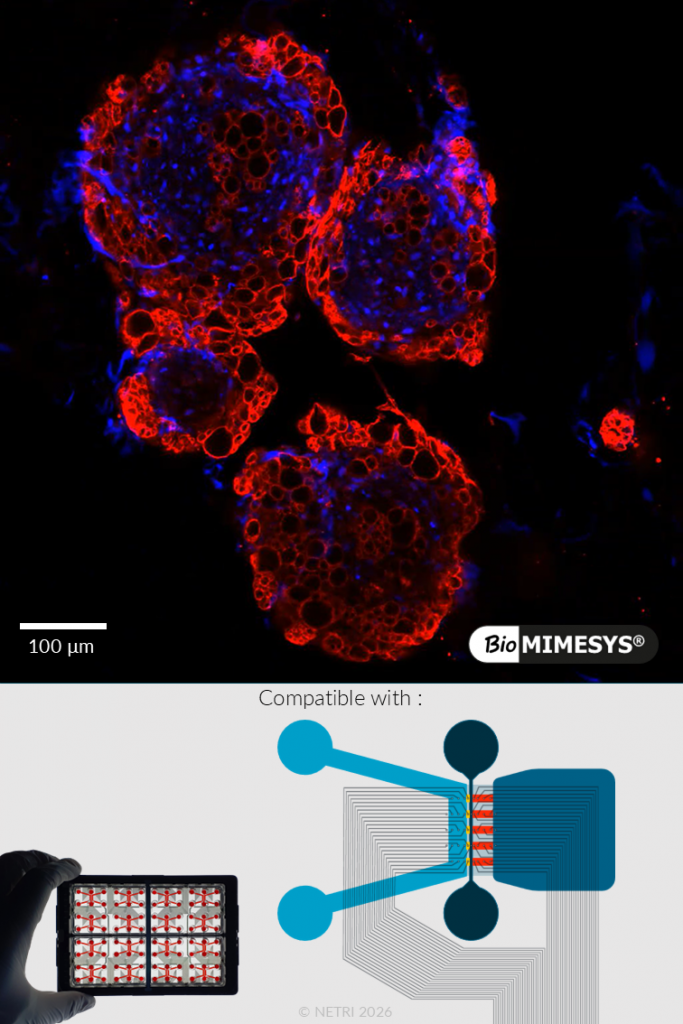
TUMOROIDS.
BioMIMESYS® enables the generation of complex 3D tumor models (tumoroids), including co-culture systems with immune cells such as macrophages. These models reproduce key features of the tumor microenvironment, including heterogeneity and cell–cell interactions.
When combined with NETRI neurofluidic MEA platforms, tumoroids can be functionally assessed through real-time electrophysiological monitoring, enabling the identification of treatment-specific signatures.
Functional validation includes: dose-dependent tumor cell viability response (e.g., 5-FU treatment), co-culture with immune cells reflecting tumor microenvironment and detection of functional treatment signatures via MEA recordings.
Key applications include:
• Oncology and tumor microenvironment modeling.
• Drug efficacy and combination therapy testing.
• Patient-derived organoid studies.
• Functional precision medicine approaches.
Available suppliers: BioMIMESYS® ONCOLOGY - ECM hydrogel.