Based on 10 years of work in academic research, NETRI was founded in June 2018 in Lyon. Considered as a TechBio, NETRI develops so-called organs-on-chip technologies that, coupled with Artificial Intelligence, predict the human effect of a drug candidate.
Located in the heart of Lyon's Gerland Biodistrict, NETRI manufactures and operates its devices in its production unit, using techniques derived from the microelectronics industry, cell culture automation and Artificial Intelligence.
•1100m² of production units.
•500m² of offices.
•35 employees: 12 PhDs within a multidisciplinary team (engineering, biology and digital).

NETRI empower pharmaceutical leaders to accelerate their drug development pipelines while maximizing return on innovation. NETRI's organs-on-chip technologies drive faster time-to-market, reduce clinical failures, and unlock new IP opportunities through repurposing and combination strategies.
By offering human-relevant models with superior predictivity and translatability, NETRI's technologies reduce reliance on non-representative animal testing, lower overall development costs, and enable earlier detection of compound limitations.
• Staying ahead of the competition.
• De-risking clinical programs.
• Being recognized as pioneers in animal-free, next-generation therapeutic innovation.
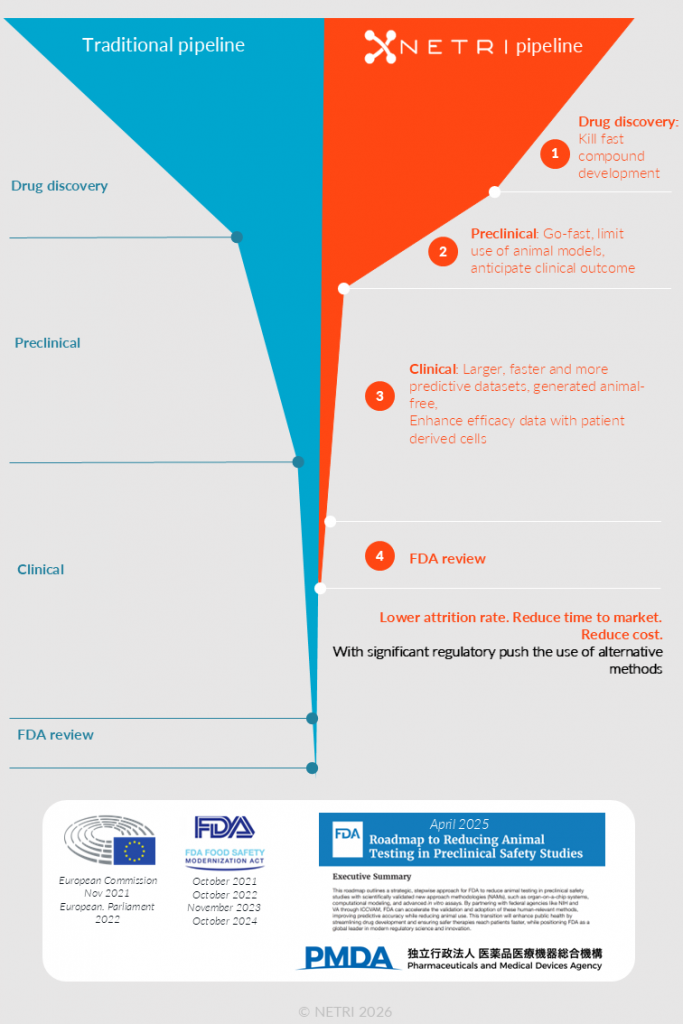
The introduction of new treatments for humans, regardless of their field of application, requires thorough testing of both toxicity and efficacy before regulatory approval.
The pharmaceutical industry is strictly overseen by authorities such as the Food and Drug Administration (FDA) in the United States, the European Medicines Agency (EMA) in the European Union, and the Agence Nationale de Sécurité du Médicament (ANSM) in France.
Today, regulatory agencies are increasingly recognizing that alternative methods can not only reduce animal testing but also offer improved predictive value and address regulatory unmet needs. They are now implementing programs and frameworks to support and qualify these approaches, providing a clear regulatory signal that human-relevant, mechanistically anchored methods are no longer just promising but becoming preferred.
NAMs are moving from the “Peak of Inflated Expectations” into the “Slope of Enlightenment,” and the time is now, with stakeholders realizing that these methods redefine safety, cost, and ethical benchmarks across the industry.
At NETRI, we believe that technological innovation must go hand in hand with strong human and societal values. Our ambition: to combine scientific excellence, respect for people, and environmental stewardship.
As a pioneer in Organs-on-Chip technologies, NETRI is committed to developing ethical, sustainable, and inclusive science to address tomorrow’s global challenges.
•Environment: Minimizing NETRI environmental footprint is a priority embedded into every aspect of NETRI operations: energy sourcing, limitation of animal testing & eco-responsible packaging and products.
•Social: NETRI is, above all, a human-driven venture led by passionate, curious, and collaborative individuals. NETRI strives to create a work environment that is inclusive, dynamic, and balanced: equal opportunity hiring, work-life balance, continuous development and societal engagement.
•Governance: NETRI decisions are driven by rigorous, transparent governance aligned with core values: ethics charter, strict compliance policy, ESG criteria and ongoing dialogue.
• 2018: NETRI founded.
• 2023: Launch of NeuroFluidics MEA (First worldwide MEA-compartmentalized) and Pain platform.
• 2025: Inauguration of France's first organ-on-a-chip production plant.
• 33 People including 12 PhDs.
NETRI PLANT.
• 1100 m² production, P2 environmental control, ISO 5 Class.
• 500 m² offices.
• 600 devices / month in 2025 & 1200 devices / month in 2026.
OF HCS PHARMA.
• Strenghten NETRI's oncological adverse events platform: adress use cases in toxicology with liver organoids.
• Key technological integration for NETRI: patent on process for preparating modified hyaluronic acid and type I collagen & patent on process for manufacturing liver organoids.
ORGANS-ON-CHIP
INITIATIVE.
• NETRI co-pilots the initiative, for France Biotech, with BioValley France: the experience of a competitiveness cluster and the field expertise of a organs on chip manufacturer.
• Structuring through an action plan to be rolled out until 2026.
INNOVATION AWARD.
• NETRI has been awarded for its pioneering NeuroFluidics Care platform.
• Transforming biochemical safety assessments, enabling early toxicity analysis of compounds used by the food industry with unprecedented precision and accessibility.
& PAIN MODEL.
• World-first compartmentalized MEA platform, the NeuroFluidics™ MEA, co-developed with Axion BioSystems, along with UpLink utility software for MEA metric evaluation.
• Pain & Injury model with a specific use case about Chemotherapy-Induced Peripheral Neuropathy.
AXOTOMY MODEL.
• NeuroFluidics™ Line, a compartmentalized organs on chip platform with three architectures devices.
• Axotomy model with DuaLink Delta Ultra.
• NeoBento high-throughput platform compatible with laboratory equipment and pumpless & standalone platform..
CHARACTERIZATION.
• Validation of five human iPSC-Derived Neurons with standard operating protocol.
• Morphological & functional characterization.
• Labelled Deeptech from Bpifrance.
NEW ARCHITECTURES
& FORMAT.
• Introduced six new architectures of organs-on-chip devices.
• Development of a high-throughput format.
& COMMERCIALIZATION.
• Set up in the competitiveness cluster Lyonbiopôle, in the Gerland biodistrict.
• First commercial unit chips.
• NETRI is founded by Thibault Honegger, CEO, and Florian Larramendy, CTO.
• Development of first-unit chips..