WETWARE.
PNS CO-CULTURE.
NETRI develops advanced Peripheral Nervous System (PNS) co-culture models designed to reproduce the functional interactions between sensory neurons and peripheral tissues. By combining human neuronal cells with relevant target tissues such as skin or muscle, these systems enable the study of neuron-tissue communication, sensory signaling and interactions in physiologically relevant in vitro environments.
These standardized co-culture models support applications in dermatology, neurobiology, and drug discovery, providing functional readouts of neuronal activation and tissue responses.
SENSORY NEURONS & KERATINOCYTES.
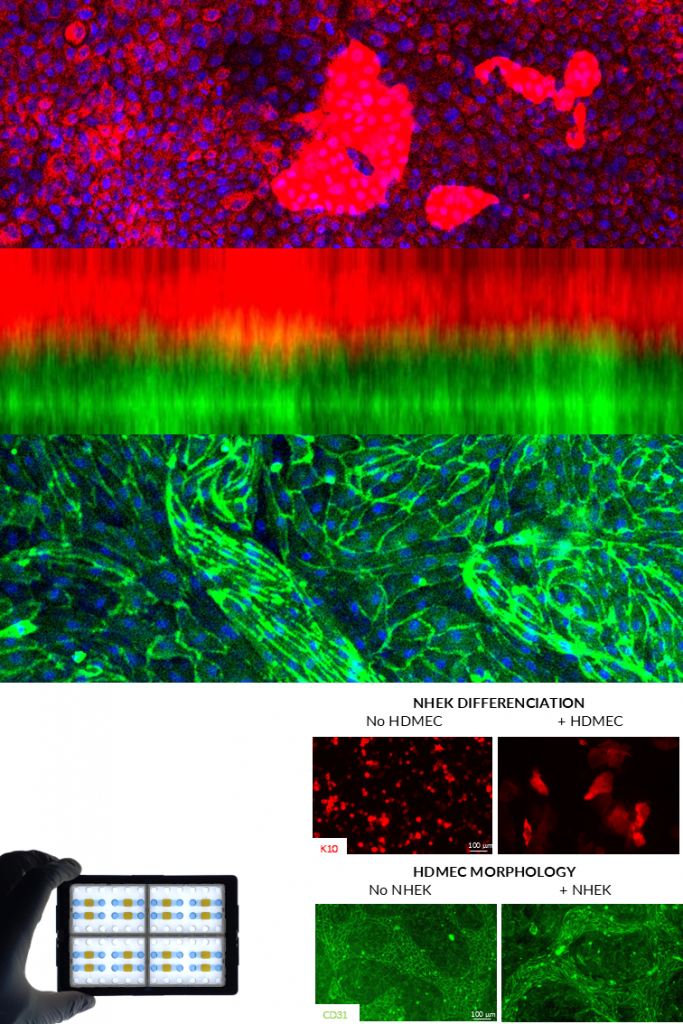
NETRI’s innervated skin model combines hiPSCs-derived sensory neurons with primary keratinocytes to reproduce key aspects of cutaneous sensory physiology. Within the compartmentalized microfluidic environment, neuronal projections extend toward the skin compartment, enabling the formation of functional neuron–tissue interfaces.
This platform enables the assessment of sensory neuron activation in response to chemical or physical stimuli in skin compartment, supporting the evaluation of compounds associated with sensations such as itching, cooling, or irritation. The model provides quantitative electrophysiological readouts of neuronal responses, enabling the study of sensory pathways relevant for dermatology and pharmaceutical applications.

KERATINOCYTES & ENDOTHELIAL CELLS.
NETRI’s vascularized skin model recreates the interaction between keratinocytes and endothelial cells to mimic the vascular component of the skin microenvironment. The two cell populations are cultured in separated compartments connected through a nanoporous membrane, allowing molecular communication while maintaining structural organization.
This configuration enables the formation of physiologically relevant cellular morphologies and differentiation patterns, including keratinocyte maturation and endothelial network organization. The model supports the evaluation of compounds targeting skin vascularization, inflammation, and barrier physiology, and can be extended to advanced 3D culture configurations.
MOTOR NEURONS & MUSCLE CELLS.
The Neuromuscular Junction (NMJ) model recreates the functional interface between motor neurons and skeletal muscle cells. In this system, hiPSC-derived motor neurons extend axons toward muscle fibers, enabling the formation of synaptic connections characteristic of neuromuscular junctions.
Morphological analyses confirm the presence of NMJ structures through markers such as acetylcholine receptor (AChR) clustering, while functional assays demonstrate muscle contraction induced by neuronal activity or pharmacological stimulation. The platform supports the study of neuromuscular physiology, synaptic function, and pharmacological modulation, providing a human-relevant model for neuromuscular diseases and drug testing.
•Publication: Towards a quality control framework for cerebral cortical organoids.
•Application Note: Traumatic Nerve Injury Platform.
•Publication: Towards a quality control framework for cerebral cortical organoids.
•Poster: Development of a brain-organoid-on-chip platform for neurotoxicity testing.
CNS CO-CULTURE.
NETRI develops advanced Central Nervous System (CNS) co-culture models that reproduce functional neuronal networks and neuron–tissue interactions in controlled microphysiological environments. Using hiPSC-derived neurons combined with compartmentalized microfluidic architectures and integrated electrophysiology, these models enable the study of neuronal connectivity, synaptic activity, and disease-relevant cellular interactions.
These standardized CNS systems provide quantitative electrophysiological readouts and support applications in neuroscience research, disease modeling, oncology, and drug discovery.
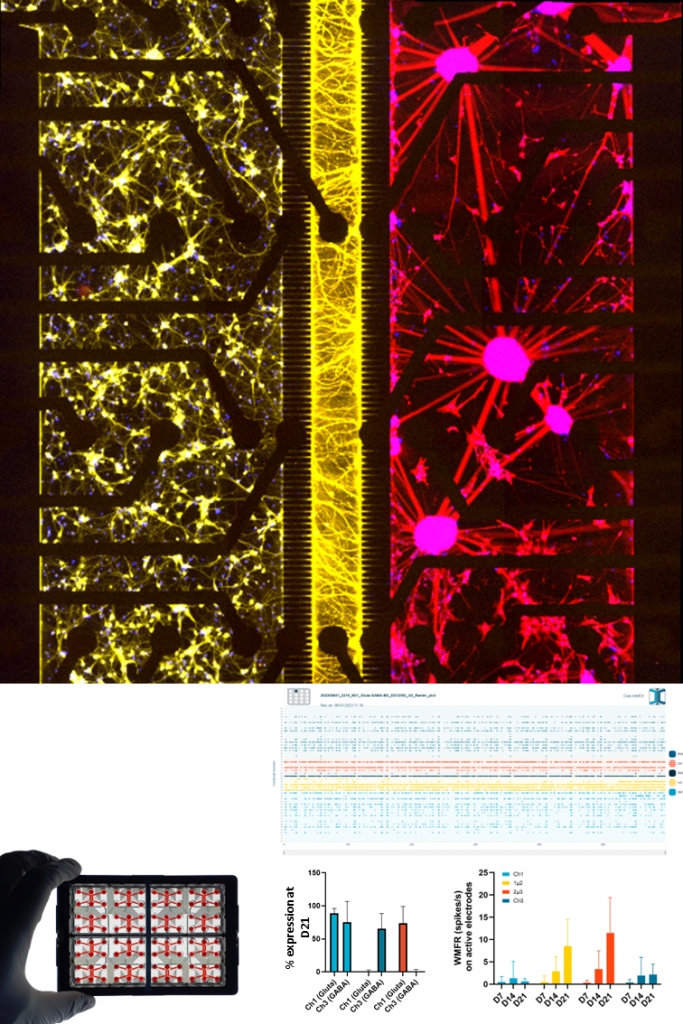
GLUTAMATERGIC & GABAERGIC NEURONS.
NETRI’s Gluta–GABA co-culture model recreates balanced neuronal networks by combining human iPSC-derived glutamatergic (excitatory) and GABAergic (inhibitory) neurons within compartmentalized microfluidic devices. This architecture allows the formation of structured neuronal circuits while enabling controlled connectivity between neuronal populations.
The system enables the monitoring of network maturation and electrophysiological activity over time using integrated microelectrode array (MEA) recordings. Functional readouts include neuronal firing rates, burst dynamics, and network synchronization, providing quantitative insights into excitatory–inhibitory balance and neuronal network function. This platform serves as a robust foundation for studying neurodevelopmental processes, neurological disorders, and pharmacological modulation of neuronal networks.
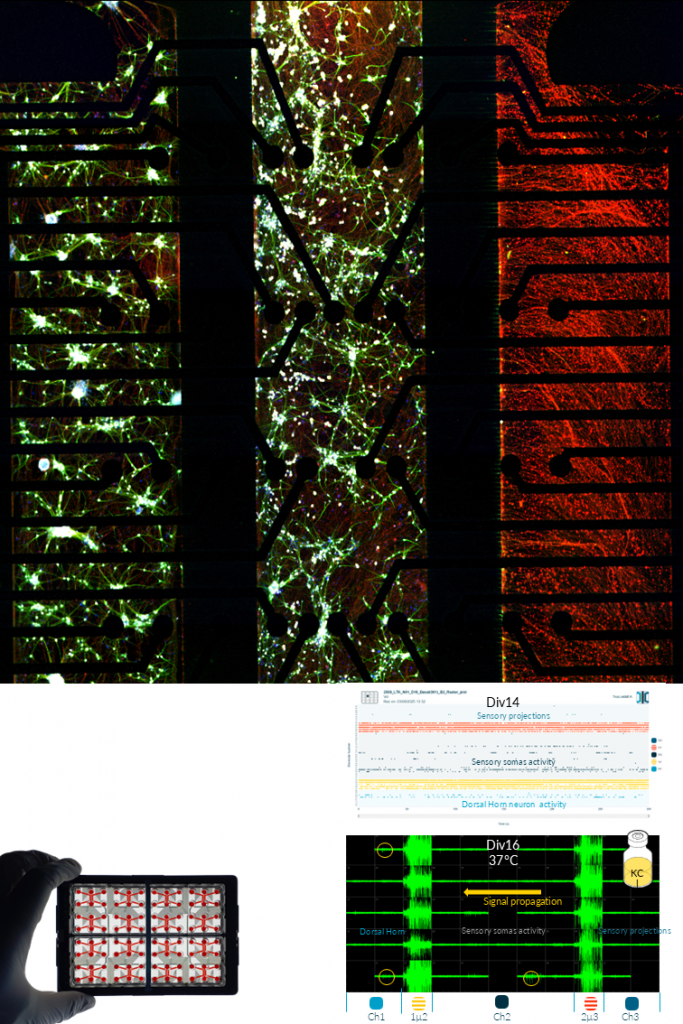
SENSORY NEURONS, ASTROCYTES & DRG.
NETRI develops advanced sensory neuron culture systems designed to reproduce functional peripheral sensory circuits relevant for pain signaling and neurotoxicity studies. These models combine human iPSC-derived sensory neurons with astrocytes, and can be extended to include dorsal horn neurons to recreate key components of the sensory–spinal pain pathway within compartmentalized microfluidic devices.
Using integrated microelectrode array (MEA) electrophysiology, these cultures enable real-time monitoring of neuronal activity, signal propagation, and network responses following pharmacological stimulation. The platform supports the generation of large electrophysiological datasets and allows the characterization of compound-specific neuronal response signatures across multiple experimental conditions.
Standardized experimental workflows enable the evaluation of drug-induced sensory effects, neurotoxicity, and inflammatory responses, supporting applications in pain research, safety pharmacology, and therapeutic compound screening. The resulting functional datasets can be further analyzed using computational clustering and mapping approaches to identify treatment-specific neuronal activity patterns.
SENSORY NEURONS & TUMOROID.
NETRI’s innervated tumoroid model combines neuronal networks with patient-derived tumor organoids to investigate the role of neuron–cancer interactions in tumor progression and treatment response. In this configuration, neuronal projections establish connections with the 3D tumor tissue, enabling the study of neuro-epithelial signaling within the tumor microenvironment.
Using NETRI’s Neuron-as-a-Sensor (NaaS) approach, neuronal electrophysiological activity can be monitored in real time to capture functional responses to therapeutic compounds. This enables label-free monitoring of treatment efficacy, tumor–neuron communication, and dynamic changes in tumor physiology. The platform supports the evaluation of anticancer therapies and combination treatments in complex 3D tumor models, providing functional treatment signatures that enhance the translational relevance of preclinical studies.

CNS CULTURE.
NETRI develops advanced human iPSC-derived CNS neuronal cultures that reproduce key neuronal phenotypes relevant to central nervous system physiology and disease. These standardized models include glutamatergic, GABAergic, dopaminergic, motor, and sensory neurons, enabling the study of specific neuronal subtypes in controlled microfluidic environments.
Combined with integrated microelectrode array (MEA) electrophysiology, these cultures enable real-time monitoring of neuronal activity, network dynamics, and pharmacological responses. These models support applications in neurological disease research, neurotoxicity assessment, and drug discovery using human-relevant neuronal systems.
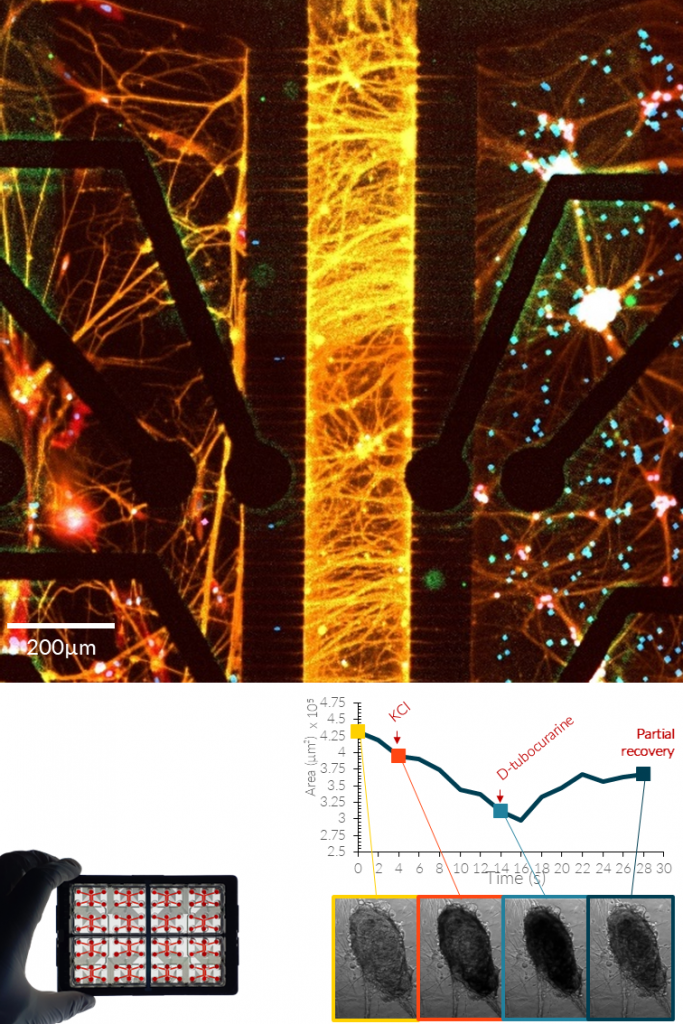
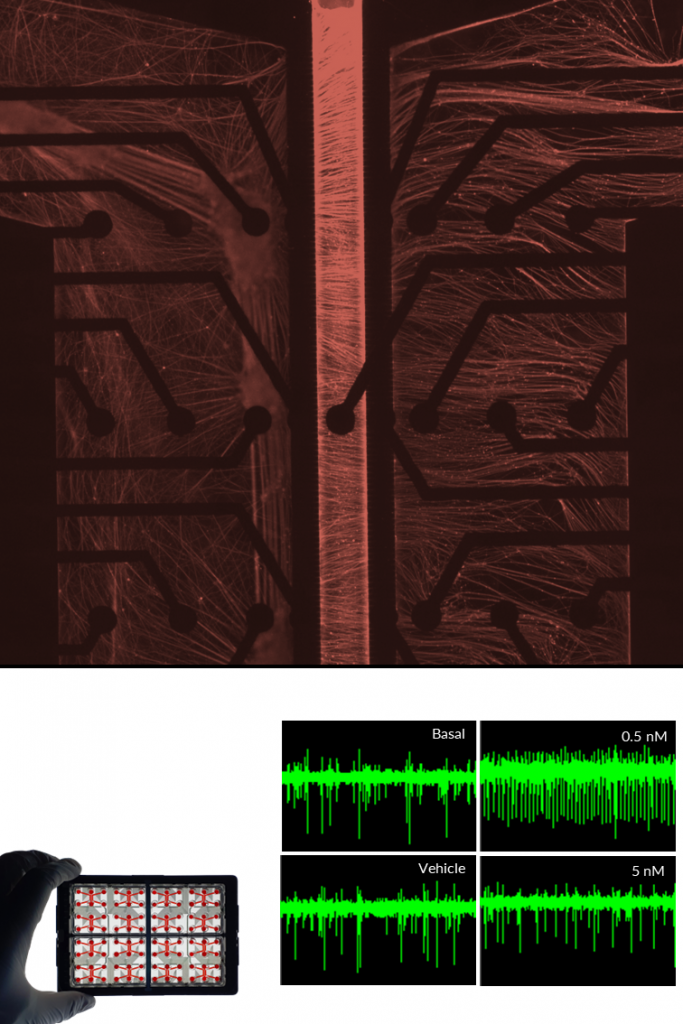
SENSORY NEURONS.
NETRI develops human iPSC-derived sensory neuron cultures that reproduce key features of peripheral sensory signaling. These neurons express characteristic sensory markers and receptors, including Nav1.7, Nav1.8, and TRPV1, enabling the study of nociceptive and mechanosensory pathways in controlled in vitro environments.
Using integrated microelectrode array (MEA) electrophysiology, the platform enables real-time monitoring of neuronal firing, stimulus-induced activity, and compound-specific responses. Sensory neurons respond to pharmacological agents and physical stimuli such as inflammatory mediators, temperature changes, osmotic stress, or ion channel modulators. These models support applications in pain research, neurotoxicity assessment, and sensory pharmacology, including the study of chemotherapy-induced peripheral neuropathy and the generation of digital functional signatures to map compound-induced sensory responses.
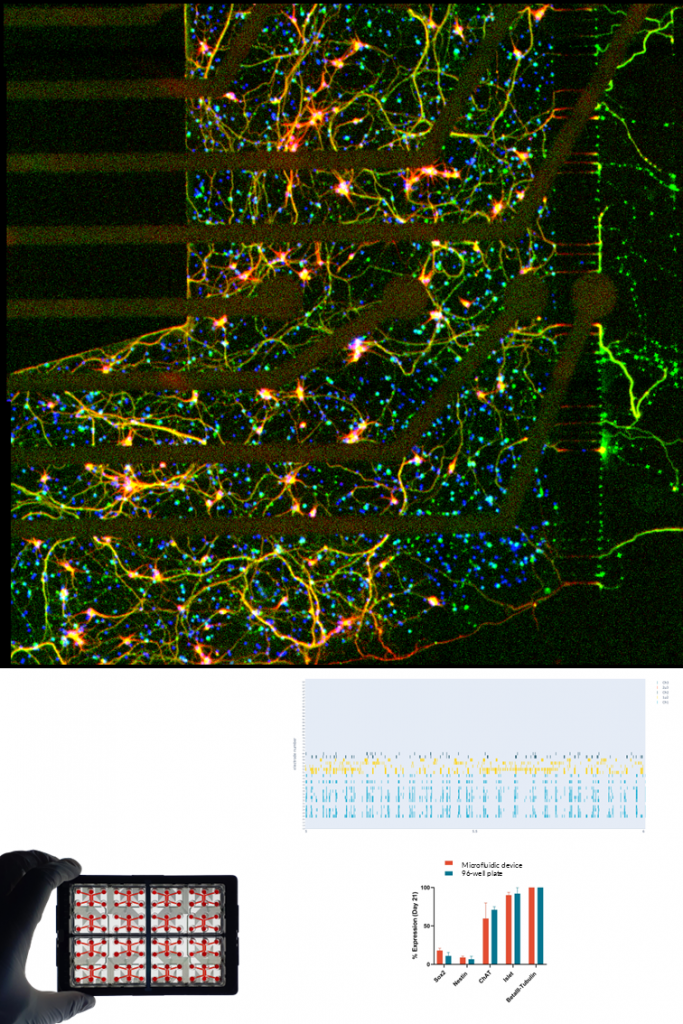
MOTOR NEURONS.
NETRI develops human iPSC-derived motor neuron cultures that form functional neuronal networks representative of motor circuitry. These neurons express characteristic motor neuron markers such as ChAT, VAChT, and Islet-1, and develop extensive axonal projections within compartmentalized microfluidic environments.
Using integrated microelectrode array (MEA) electrophysiology, the platform enables real-time monitoring of neuronal activity, network dynamics, and axonal signal propagation. The compartmentalized architecture also allows the study of axon injury and regeneration processes by selectively applying compounds to specific neuronal compartments. These models support applications in motor neuron disease research, neurotoxicity assessment, and regenerative neuroscience, providing quantitative measurements of axon growth, degeneration, and regrowth dynamics following pharmacological or physical perturbations.
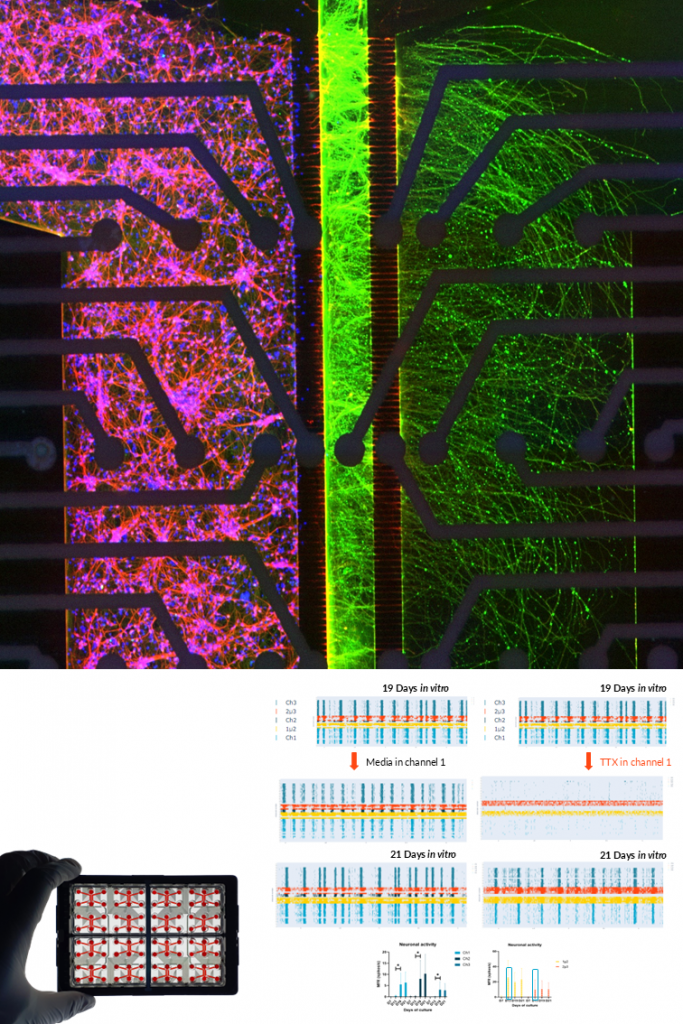
GLUTAMATERGIC NEURONS.
NETRI develops human iPSC-derived glutamatergic neuron cultures that form functional excitatory neuronal networks within compartmentalized microfluidic devices. These neurons exhibit mature neuronal morphology and spontaneous electrophysiological activity, enabling the study of excitatory signaling in controlled in vitro environments.
Using integrated microelectrode array (MEA) electrophysiology, the platform enables real-time monitoring of neuronal firing, network activity, and pharmacological responses. These models support applications in neurological disease research, neurotoxicity assessment, and drug discovery, including the detection of disease-related functional signatures in human neuronal networks.
DOPAMINERGIC NEURONS.
NETRI develops human iPSC-derived dopaminergic neuron cultures that reproduce key features of midbrain dopaminergic networks relevant to neurological disease research. These neurons exhibit characteristic dopaminergic markers and develop functional neuronal networks with spontaneous electrophysiological activity within compartmentalized microfluidic devices.
Using integrated microelectrode array (MEA) electrophysiology, the platform enables real-time monitoring of neuronal firing, network activity, and pharmacological modulation. These models support applications in neurodegenerative disease research, particularly Parkinson’s disease, as well as drug discovery and neurotoxicity assessment targeting dopaminergic pathways.
GABAERGIC NEURONS.
NETRI develops human iPSC-derived GABAergic neuron cultures that model inhibitory neuronal networks within controlled microfluidic environments. These neurons establish functional inhibitory circuits that play a critical role in regulating neuronal network balance and synaptic activity.
Using integrated microelectrode array (MEA) electrophysiology, the platform enables the monitoring of neuronal firing patterns, inhibitory signaling, and compound-induced modulation of network activity. These models support applications in neurological disorder research, neuropharmacology, and safety pharmacology, particularly for studying mechanisms related to neuronal excitability and inhibitory network regulation.