MICROFLUIDICS IS NOW JUST
1-CLICK AWAY WITH NETRI SHOP
Discover our new exclusive package
organs-on-chip kits and all our
neuro-organs-on-chip devices.

The skin is a protective organ, able to decode a wide range of tactile, thermal, or noxious stimuli. Psoriasis, atopic dermatitis, eczema, wound healing, skin irritation, herpes infection, hyperhidrosis, neuropathy, and skin sensitivity syndrome are conditions involving both skin cells, i.e., keratinocytes and neurons. Preclinical research is limited by in vitro models, which are not always relevant either because they often use rodent neurons cells or because the cocultures of skin cells and neurons do not fully recapitulate the anatomical structure, i.e. , a neuronal cell body compartmentalized from the skin.
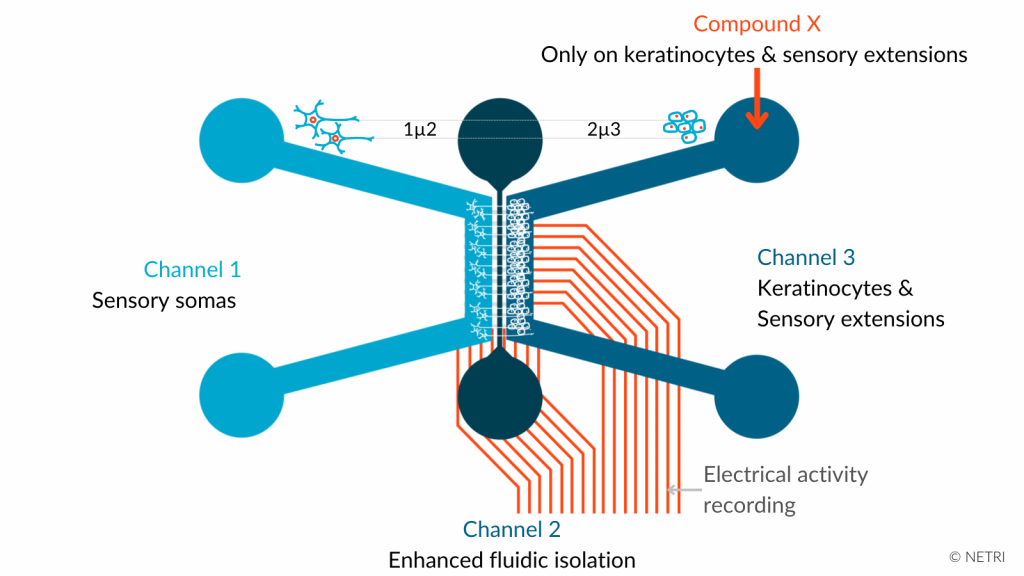
The association between compartmentalized microfluidic device technology and human-derived sensory neurons may address these limitations. The interest in this model is to (i) apply a stimulus on the skin and evaluate the impact on neurons or (ii) apply a stimulus on neurons and evaluate the impact on the skin.
• DuaLink NeuroFluidics Devices & Skin NeuroFluidics Cultures
• Primary keratinocytes in channel 3
• hiPSC-derived sensory neurons somas in channel 1 & extensions in channel 3
• Compound response in channel 3
This Use Case shows that, by using neurons as bio-digital sensors, the electrical effects of a compound applied to the healthy skin compartment can be recorded on neurons. This opens the way to screening compounds on altered models (sensitive skin, atopic dermatosis...).
• Evaluation of the effect on neuron electrical activity following treatment
• Acute and « chronic »activity modulation
• Comparison with compounds of reference
Test your compound on our ready-to-use NeuroFluidics Cultures with reference compound already on the market or having failed in the clinical phase.

Cosmetic regulation ensures product safety, labeling accuracy, and adherence to quality standards in the beauty industry.
• No animal testing
• Drug and cosmetic efficacy and toxicity
• Compatible with standard analytical method and tools
Compartmentalized microfluidics devices to recreate skin compartment and neurons or endothelial cells compartment.
• Discrimination of mode and mechanism of action
• Pathophysiological applications (sensitive skin and pruritus, inflammatory dermatosis…)
NeoBento™, the standard SBS format for NeuroFluidics Devices for readouts and lab equipments compatibility.
• Pump-free & expensive equipment-free
• Standard equipment compatibility
• Electrophysiology, imaging and biochimic analysisreadouts compatibility
Soft silicon material, optically transparent for microscopy compatiblity and porous gas permability
• Imaging (Immunofluorescence, Calcium Imaging…)
• Confocal Imaging
• Perfusion-free with high biocompatibility
• Technological Transfert
• FTE & Screening Services
• Co-development
• Analytical Services
• NeuroFluidics Devices
• NeuroFluidics Cultures
• NeuroFluidics Digital
• Training & Organs-on-chip Kits
Discover our new exclusive package
organs-on-chip kits and all our
neuro-organs-on-chip devices.

ORGANS-ON-CHIP KITS
Quickly and easily adopt organs-on-chip
into users’ research