2-Cell Culture MEA Electrophysiology NEUROFLUIDICS™ LINE
DUALINK
Co-culture & fluidic isolation
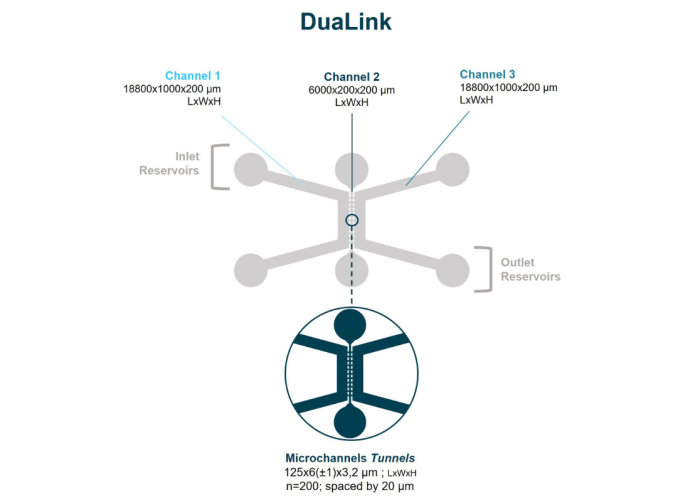
With the DuaLink microfluidic chip, microchannels tunnels and HT format NeoBento technologies are combined in a single chip to provide an unparalleled user experience combined with an enhanced fluidic isolation.
- Co-culture of different cell types
- Compartmentalization
- Fluidic isolation
- Discontinious connectivity by microchannels tunnels technology
- 3 compartments : 2 for cell culture & 1 for fluidic isolation
- 8 or 16 microfluidic chips per NeoBento
- • Human cells
- • Rodent cells
- • Immunofluorescence
- • Live Dead Assays
- • Live staining
- • Liquid chromatography
- • Mass Spectoscopy
- • Lysis cells/supernatant analysis
- • ELISA
- • Calcium Imaging
- • Drug screening
- • Quantitative assays
- • Axonal transport
- • Skin nociception
- • Itch
- • Ageing
- • Wound healing
- • Toxicology
- • Virology (viral transfection in one compartment only)
- • Neuroinflammation (Multiple sclerosis, Cerebral tumors...)
- • Innervated skin
- • Neuromuscular junction
- • Motor neuron diseases (Amyotrophic Lateral Sclerosis...)
•
Microchannels tunnels: Microchannels tunnels are rectangular slits engraved within microfluidic chips. Due to their micrometric measurements (3µm height, 5µm width), only cell extensions like for example neurites can grow within the microchannels, leaving the cell bodies within the compartments themselves. They allow also an enhanced fluidic isolation and discontinuous connectivity.
• NeoBento™: The NeoBento™ format makes NETRI’s microfluidic devices as versatile and easy to handle as a typical 96-well plate, by being in the standard AINSI format. The NeoBento™ is compatible with routine control magnification imaging (x10, x20,...) and confocal or high magnification (x63 with water immersion) imaging. Its versatility allows users to perform High Throughput Screening, histochemical analysis and individual cell fixation. Finally, its compounds permit an optimized oxygenation without expensive equipment (pump or stirrer) and a controlled humidity of cell culture.
• NeoBento™: The NeoBento™ format makes NETRI’s microfluidic devices as versatile and easy to handle as a typical 96-well plate, by being in the standard AINSI format. The NeoBento™ is compatible with routine control magnification imaging (x10, x20,...) and confocal or high magnification (x63 with water immersion) imaging. Its versatility allows users to perform High Throughput Screening, histochemical analysis and individual cell fixation. Finally, its compounds permit an optimized oxygenation without expensive equipment (pump or stirrer) and a controlled humidity of cell culture.
Publications
Microchannel patterning strategies for in vitro structural connectivity modulation of neural networks
Electrokinetic confinement of axonal growth for dynamically configurable neural networks
Microfluidic neurite guidance to study structure-function relationships in topologically complex population-based neural networks
Microfluidic Devices Protocols
NeuroFluidics™ – DuaLink - DuaLink MEA
Cells Types Protocols
Sensory Neurons – Axol Bioscience
Application Notes
Evaluation of amyloid beta oligomers (AβO) effects on functional network integrity of rodent hippocampal neurons
Posters
Microfluidic high-throughput screening platform to screen pre-clinical stage compound effects on neurite outgrowth of human Motor Neurons post-injury
Standardization criteria of hiPSC-derived neurons for Brain-on-Chip applications
Modeling the human Brain-on-Chip with human iPSC-derived Glutamatergic neurons
Standardization criteria of hiPSC-derived neurons for Brain-on-Chip applications
Microchannel patterning strategies for in vitro structural connectivity modulation of neural networks
Electrokinetic confinement of axonal growth for dynamically configurable neural networks
Microfluidic neurite guidance to study structure-function relationships in topologically complex population-based neural networks
Microfluidic Devices Protocols
NeuroFluidics™ – DuaLink - DuaLink MEA
Cells Types Protocols
Sensory Neurons – Axol Bioscience
Application Notes
Evaluation of amyloid beta oligomers (AβO) effects on functional network integrity of rodent hippocampal neurons
Posters
Microfluidic high-throughput screening platform to screen pre-clinical stage compound effects on neurite outgrowth of human Motor Neurons post-injury
Standardization criteria of hiPSC-derived neurons for Brain-on-Chip applications
Modeling the human Brain-on-Chip with human iPSC-derived Glutamatergic neurons
Standardization criteria of hiPSC-derived neurons for Brain-on-Chip applications