MICROFLUIDICS IS NOW JUST
1-CLICK AWAY WITH NETRI SHOP
Discover our new exclusive package
organs-on-chip kits and all our
neuro-organs-on-chip devices.


• DuaLink, DuaLink MEA or DuaLink Shift NeuroFluidics Devices
• Duplex Well NeuroFluidics Devices
• hiPSC Neurons / Rodent Neurons / Organoid
• Microchannels technology to segregate mode of action of compound and co-culture different cell types
CNS Models enable the creation of an in vitro compartmentalized model mimicking human anatomy. This enables also to co-culture neuronal and non-neuronal cell types (Neurons & Tumor Cells, Neurons & Immune Cells…). By adding the MEA option, evaluate the functional impact of compound on neurons.
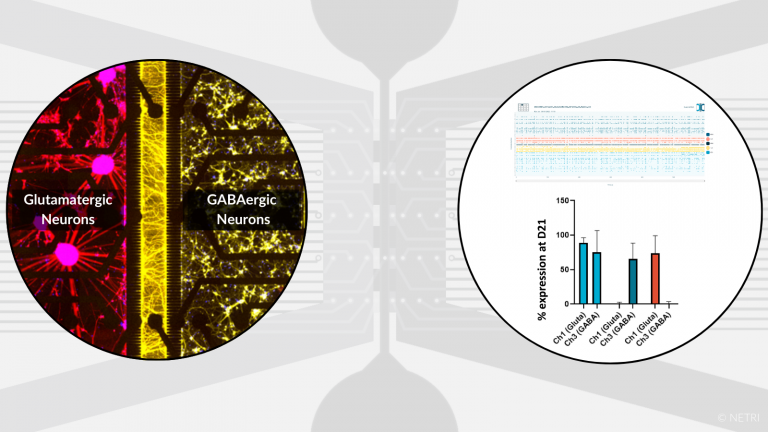
Co-culture of hiPSC-derived glutamatergic neurons and GABAergic neurons for Central Nervous System applications.
• DuaLink or DuaLink MEA for screening of drug candidate
• DuaLink Shift MEA for synapses electrophysology activity isolation per compartment
• Electrophysiology, Imaging and Biochimis analysis readouts
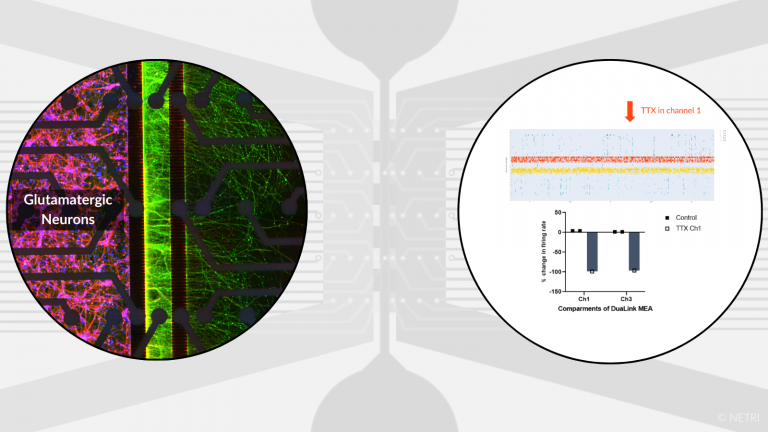
Co-culture of hiPSC-derived glutamatergic neurons with non-neuronal cells (tumor, immune…) for oncology and immunology applications.
• DuaLink MEA for screening of drug candidate
• DuaLink Shift MEA for synapses electrophysology activity isolation per compartment
• Electrophysiology, Imaging and Biochimis analysis readouts
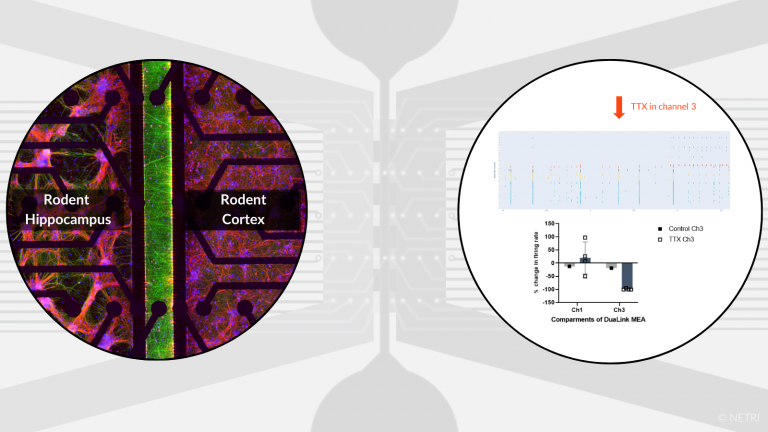
Co-culture of rodent cortical & hippocampal neurons for neurological dosorders applications.
•
DuaLink MEA to evaluate the functional impact of compound on neurons and for screening of drug candidate
• Electrophysiology, Imaging and Biochimis analysis readouts
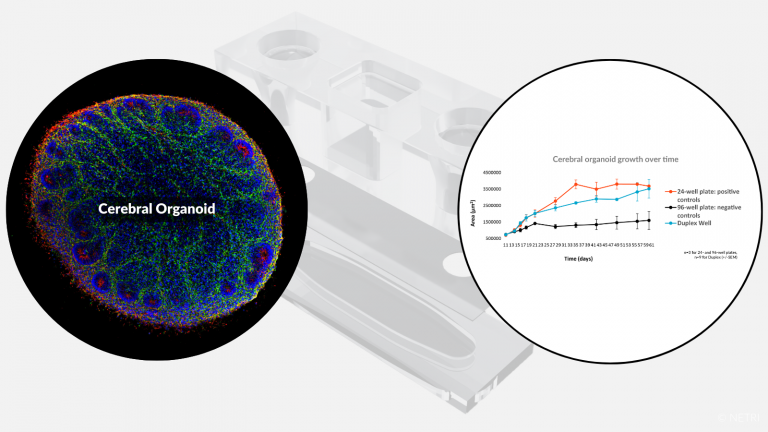
Culture of cerebral organoid up to 60 days in vitro for preclinical applications.
• Duplex Well to perfuse cerebral organoid
• Standard protocol for growing organoids-on-chip
• Characterization of brain organoid-on-chip
NeoBento™, the standard format for NeuroFluidics Devices chips, available up to 4 QuarterBentos™ (up to 16 chips).
• Standard ANSI format (96-well plate)
• Pump-free & expensive equipment-free
• Standard equipment (liquid handling & imaging) compatibility
Humanized in vitro CNS models for a wide variety of applications.
• Mimick human anatomy
• Up to 2 separated but connected neurons cell type
• Screening of potential drugs or therapeutic interventions for their effectiveness in preventing or treating therapies
In-depth reading of the data to better understand the study results and potential implications.
• Electrophysiological recording (MEA)
• Imaging (Immunofluorescence, Calcium Imaging…)
• Biochimic analysis (ELISA, Lysis cells analysis, Liquid Chromatography…)
Compartmented microfluidic devices for applying compounds specifically to a cell compartment.
• Discrimination of mode of action and mechanism of pathology
• Segregation of therapeutic modality: topical versus systemic
• Technological Transfert
• FTE & Screening Services
• Co-development
• Analytical Services
• 3 architectures with or without MEA-recording
• 8 or 16 data points per plate
• Training
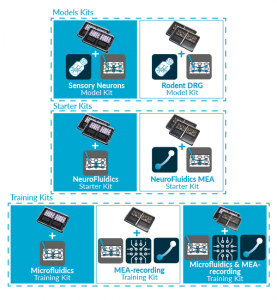
• Organs-on-chip Kits
Discover our new exclusive package
organs-on-chip kits and all our
neuro-organs-on-chip devices.

ORGANS-ON-CHIP KITS
Quickly and easily adopt organs-on-chip
into users’ research