MICROFLUIDICS IS NOW JUST
1-CLICK AWAY WITH NETRI SHOP
Discover our new exclusive package
organs-on-chip kits and all our
neuro-organs-on-chip devices.

Inflammatory bowel diseases (IBD) are complex chronic inflammatory disorders of the gastrointestinal (GI) tract. Recent evidence suggests that the gut-brain axis may be pivotal in gastrointestinal and neurological diseases, especially IBD. Modeling the gut-brain axis involved in IBD, which involves bilateral communication of neuronal, hormonal, metabolic, immunological and microbial signals, remains a challenge due to its high biological complexity, the involvement of different cell types and physical communication pathways, and the lack of study models. These include animal models, which do not allow for the study of the pathophysiological effects of IBD on mucosal nerve cells, and gut-on-chip models, which do not incorporate nerve cells and analysis of their electrical activity (MEA).
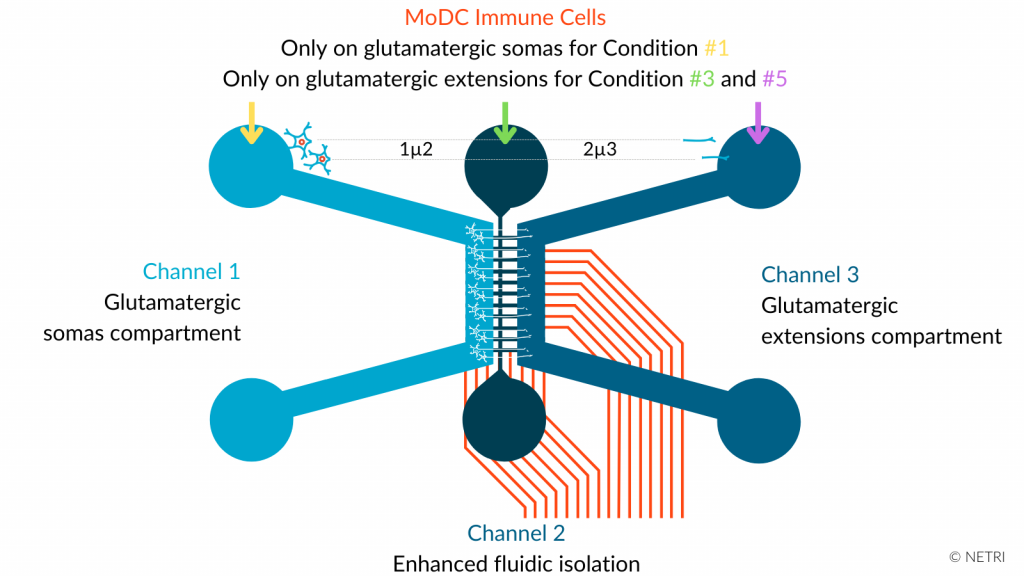
Modeling the gut-brain axis and studying neuron-immune cell communication. The DuaLink Shift comprises three compartments with an asymmetric channel, enabling soma and neurites to be isolated by microchannels to create synaptic transmission. The microfluidic device was coupled to MEA to record the electrophysiological signal as a function of conditions and analyze neurite functionality.
• DuaLink Shift MEA NeuroFluidics Devices & CNS NeuroFluidics Cultures
• hiPSC-derived glutamatergic neurons in Channel 1
• MoDC in either Channel 1, Channel 2 or Channel 3 depending on conditions
• Analyze of the electrical impact of MoDC cells on human glutamatergic neurons
The use case demonstrates (i) the association of human neuronal and immune cells in DuaLink Shift and (ii) access to a non-invasive analysis method to study the communication between these two cell types in the context of inflammation to model neuro-immune interaction in the inflammatory condition of IBD.
• Observation of IL-6 secretion induced by the addition of MoDCs
• Induction of changes in the electrical activity of glutamatergic neurons by the addition of MoDCs
• Creation of a specific model of neuron-immune cell communication with potential implications for understanding the impact of immunological signaling pathways on neuronal activity
Test your compound on our ready-to-use NeuroFluidics Cultures with reference compound already on the market or having failed in the clinical phase.

The study explores gut-brain communication in IBDs, focusing on neuroimmune interactions using a microfluidic device.
• A new approach coupled with MEA to model the neuro-immune system
• Communication of neuronal, hormonal, metabolic, immunological and microbial signals
NeoBento™, the standard format for NeuroFluidics Devices chips, available up to 4 QuarterBentos™ (up to 16 chips).
• Standard ANSI format (96-well plate)
• Pump-free & expensive equipment-free
• Standard equipment (liquid handling & imaging) compatibility
This device allows first the precise deposition of cells in a chamber directly on MEA.
• Co-culture and connection of neurons & immune cells
• Control of cell seeding density and homogeneity on MEA
• Control of media exchange, which is a critical step for human neural progenitor differentiation directly into devices
In-depth reading of the data to better understand the study results and potential implications.
• Electrophysiological recording (MEA)
• Imaging (Immunofluorescence, Calcium Imaging…)
.
• Biochimic analysis (ELISA, Lysis cells analysis, Liquid Chromatography…)
• Technological Transfert
• FTE & Screening Services
• Co-development
• Analytical Services
• NeuroFluidics Devices
• NeuroFluidics Cultures
• NeuroFluidics Digital
• Training & Organs-on-chip Kits
Discover our new exclusive package
organs-on-chip kits and all our
neuro-organs-on-chip devices.

ORGANS-ON-CHIP KITS
Quickly and easily adopt organs-on-chip
into users’ research