MICROFLUIDICS IS NOW JUST
1-CLICK AWAY WITH NETRI SHOP
Discover our new exclusive package
organs-on-chip kits and all our
neuro-organs-on-chip devices.

Pediatric high-grade gliomas (pHGG) represent childhood and adolescent brain cancers that carry a rapid dismal prognosis. Since there is a need to overcome the resistance to current treatments and find a new way of cure, modeling the disease as close as possible in an in vitro setting to test new drugs and therapeutic procedures is highly demanding. Studying their fundamental pathobiological processes, including glutamatergic neuron hyperexcitability, will be a real advance in understanding interactions between the environmental brain and pHGG cells.
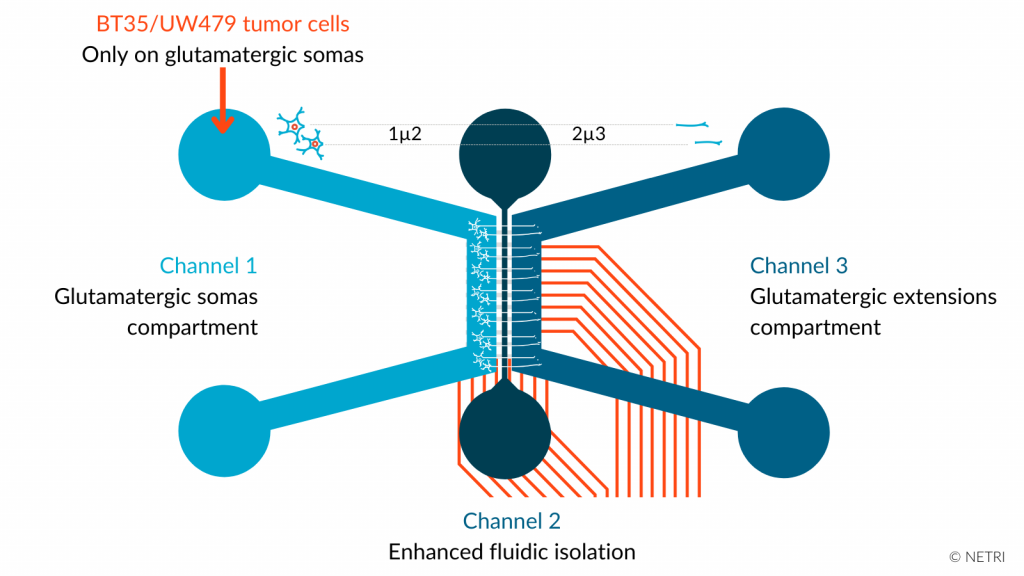
Organs-on-chip enable to recreate neurons/pHGG cell interactions with the development of a functional in vitro model co-culturing human-induced Pluripotent Stem (hiPS)-derived cortical glutamatergic neurons and pHGG cells and recording their electrophysiological modifications.
• DuaLink NeuroFluidics Devices & CNS NeuroFluidics Cultures
• hiPSC-derived glutamatergic neurons in Channel 1
• BT35, patient-derived cell line, or, UW479, commercialized pHGG line, in Channel 1
• Analyze of the electrical impact of pHGG cells on human glutamatergic neurons
This Use Case shows the development of a functional in vitro co-culture model of human induced pluripotent stem cell (hiPS)-derived cortical glutamatergic neurons and pHGG-derived cell lines in compartmentalized microfluidic devices, and a process for recording electrophysiological changes in glutamatergic neurons.
• Differentiation and characterization of hiPSC-derived glutamatergic neurons (Nestin, Sox2, mGlurR2 & vGLUT1)
• Analysis of the electrical impact of pHGG cells on glutamatergic neurons
• Significant increase in the excitability of glutamatergic neurons in the presence of tumor cells
Test your compound on our ready-to-use NeuroFluidics Cultures with reference compound already on the market or having failed in the clinical phase.

The study shows the development of a functional in vitro model co-culturing human neurons and tumor cells into compartmentalized microfluidic devices and a process to record their electrophysiological modifications.
• Screening of potential drugs for their effectiveness in treating Pediatric Glioblastoma
NeoBento™, the standard format for NeuroFluidics Devices chips, available up to 4 QuarterBentos™ (up to 16 chips).
• Standard ANSI format (96-well plate)
• Pump-free & expensive equipment-free
• Standard equipment (liquid handling & imaging) compatibility
Recreate the brain microenvironment to co-culture neurons and tumor cells.
• Co-culture and connection of neurons & immune cells
• Control of media exchange and cell seeding density
• Analysis of the electrical impact of tumor cells on neurons
In-depth reading of the data to better understand the study results and potential implications.
• Electrophysiological recording (MEA)
• Imaging (Immunofluorescence, Calcium Imaging…)
.
• Biochimic analysis (ELISA, Lysis cells analysis, Liquid Chromatography…)
• Technological Transfert
• FTE & Screening Services
• Co-development
• Analytical Services
• NeuroFluidics Devices
• NeuroFluidics Cultures
• NeuroFluidics Digital
• Training & Organs-on-chip Kits
• DuaLink MEA
Discover our new exclusive package
organs-on-chip kits and all our
neuro-organs-on-chip devices.

ORGANS-ON-CHIP KITS
Quickly and easily adopt organs-on-chip
into users’ research